Fenbendazole has attracted remarkable interest as a repurposed antiparasitic with potential anticancer activity. For most people using standard doses, its safety profile is reassuring. Yet as off-label use has expanded, a small but important body of peer-reviewed case reports has documented serious drug-induced liver injury (DILI) in a subset of patients — particularly those who escalate doses rapidly or combine fenbendazole with immune checkpoint inhibitor (ICI) therapies. Understanding these cases is essential for anyone considering fenbendazole as part of a cancer protocol.
This article reviews all three published case reports of fenbendazole-associated hepatotoxicity, examines the biological mechanisms behind liver injury, and provides a practical monitoring framework. It also covers liver-protective supplements, the rationale behind scheduled dosing breaks, and clear guidance on who should avoid fenbendazole entirely. The goal is not to alarm — the overwhelming majority of people at standard doses have no liver problems — but to give you the information you need to use this compound as safely as possible.
⚠️ Educational Disclaimer
This article is for research and informational purposes only. It does not constitute medical advice and is not a substitute for professional oncology consultation. Always discuss any investigational protocol with your physician.
🔬 How Fenbendazole Is Processed by the Liver
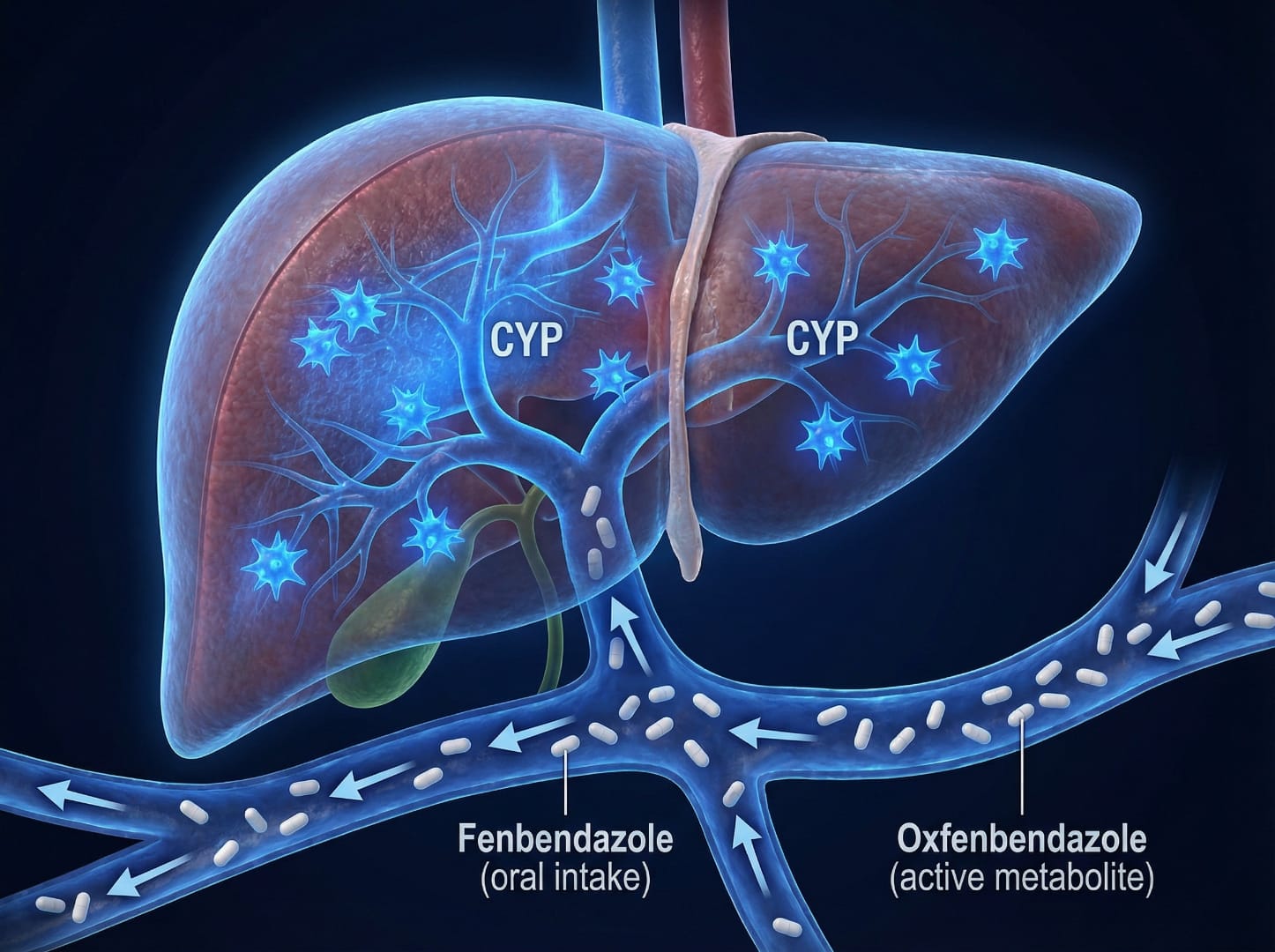
Fenbendazole is extensively metabolized by the liver through first-pass hepatic processing, converted primarily into its active metabolite fenbendazole sulfoxide (oxfendazole) via cytochrome P450 enzymes, particularly CYP1A1 and CYP1A2. Under normal circumstances this process is efficient and well-tolerated. At elevated doses or with rapid dose escalation, several mechanisms can overwhelm hepatic capacity:
- Glutathione depletion: FBZ metabolism generates reactive oxygen species. Glutathione is the liver's primary antioxidant defense; at high metabolic loads, these stores become depleted, leaving hepatocytes vulnerable to oxidative damage.
- CYP1A1/CYP1A2 over-activation: Excessive enzymatic throughput generates toxic intermediate metabolites that can directly damage hepatocyte membranes and mitochondria.
- Microtubule disruption in hepatocytes: FBZ's core mechanism — inhibiting tubulin polymerization — is not selective to cancer cells. At higher systemic exposures, it can impair hepatocyte cytoskeletal architecture and bile secretion.
- Immune-mediated amplification: Combined use with immune checkpoint inhibitors (anti-PD-1, anti-LAG-3) can amplify the hepatic inflammatory response, potentially worsening DILI severity.
These mechanisms are dose-dependent and exposure-dependent — clinically significant at doses substantially above the standard 222 mg three times per week, or when daily dosing is sustained without breaks.
🩺 Published Case Reports: A Detailed Review
Case 1: Dose Escalation in Metastatic Colon Cancer (2026)
A 2026 paper in the World Journal of Clinical Cases (PMC12836008) reports a 47-year-old woman with Lynch syndrome-associated metastatic colon cancer on nivolumab/relatlimab (Opdualag). After nine months on ICI therapy without liver problems, she began fenbendazole at 222 mg three times per week — consistent with the Joe Tippens Protocol — tolerated for ~6 weeks. She then escalated to 222 mg daily, a 2.3-fold increase in weekly exposure (666 mg → 1,554 mg/week). Within 7 days: AST 2,435 U/L (~61× ULN), ALT 2,407 U/L (~60× ULN), total bilirubin 3.1 mg/dL, R ratio 11.2 (hepatocellular pattern). RUCAM score: 8 (probable causality). The last ICI dose had been 4 weeks prior, ruling out immune-mediated hepatitis. Fenbendazole was discontinued; ALT fell 78% within 10 days. Immunotherapy was safely resumed one month later.
📌 Key insight: A 2.3× increase in weekly exposure — switching from 3×/week to daily — triggered severe hepatotoxicity within a week.
Case 2: Histologically Confirmed Severe DILI (2024)
An ACG Case Reports Journal paper (PMC11068125) describes a 67-year-old woman who had self-administered three 1-gram packets of fenbendazole three times per week (~9 g/week) for approximately one year based on social media anticancer claims. She presented with AST 1,869 U/L, ALT 2,600 U/L, total bilirubin peaking at 24 mg/dL, INR 1.6 — meeting DILIN criteria for severe DILI. Liver biopsy confirmed centrilobular hepatocyte necrosis with lymphocyte-predominant portal infiltrate. RUCAM score: 9 (highly probable) — the highest reported in any published FBZ case. Despite severity, she recovered with supportive care alone; all LFTs normalized within three months of stopping fenbendazole.
📌 Key insight: Biopsy-confirmed DILI after one year of high-dose use (~9 g/week). Fully reversible after cessation — no permanent damage.
Case 3: NSCLC Patient on Pembrolizumab (2021)
The earliest case, in Case Reports in Oncology (PMC8255718), involved an 80-year-old woman with advanced NSCLC on pembrolizumab monotherapy. She independently began fenbendazole 1 g on a 3-on/4-off schedule (4.5× the standard dose). After ~1 month, routine monitoring found AST elevated from 24 to 386 U/L, ALT from 16 to 487 U/L — the patient was asymptomatic. Naranjo score: 6 (probable). FBZ was stopped; LFTs normalized within ~7 weeks. Pembrolizumab was restarted without recurrence. Notably, no tumor shrinkage was observed — CEA rose during the FBZ period.
📌 Key insight: Even 3-on/4-off schedules can cause injury at 1 g doses. Asymptomatic injury was caught only by routine monitoring — underscoring its importance.
📊 Summary Comparison of All Three Cases
| Feature | Case 1 (2026) | Case 2 (2024) | Case 3 (2021) |
|---|---|---|---|
| Patient | 47yo woman | 67yo woman | 80yo woman |
| Cancer | Metastatic colon (Lynch) | Prior colon ca / skin lesion | NSCLC stage IVB |
| FBZ Dose (trigger) | 222 mg/day (escalated) | 3 g × 3×/wk (~9 g/wk) | 1 g × 3-on/4-off |
| Concurrent ICI | Yes (nivolumab/relatlimab) | No | Yes (pembrolizumab) |
| Peak AST / ALT | 2,435 / 2,407 U/L | 1,869 / 2,600 U/L | 386 / 487 U/L |
| RUCAM/Naranjo | RUCAM 8 — probable | RUCAM 9 — highly probable | Naranjo 6 — probable |
| Biopsy | No | Yes — confirmed DILI | No |
| Resolution | ~10 days (78% ALT drop) | 3 months (full LFT normalization) | ~7 weeks (full LFT normalization) |
| Source | PMC12836008 | PMC11068125 | PMC8255718 |
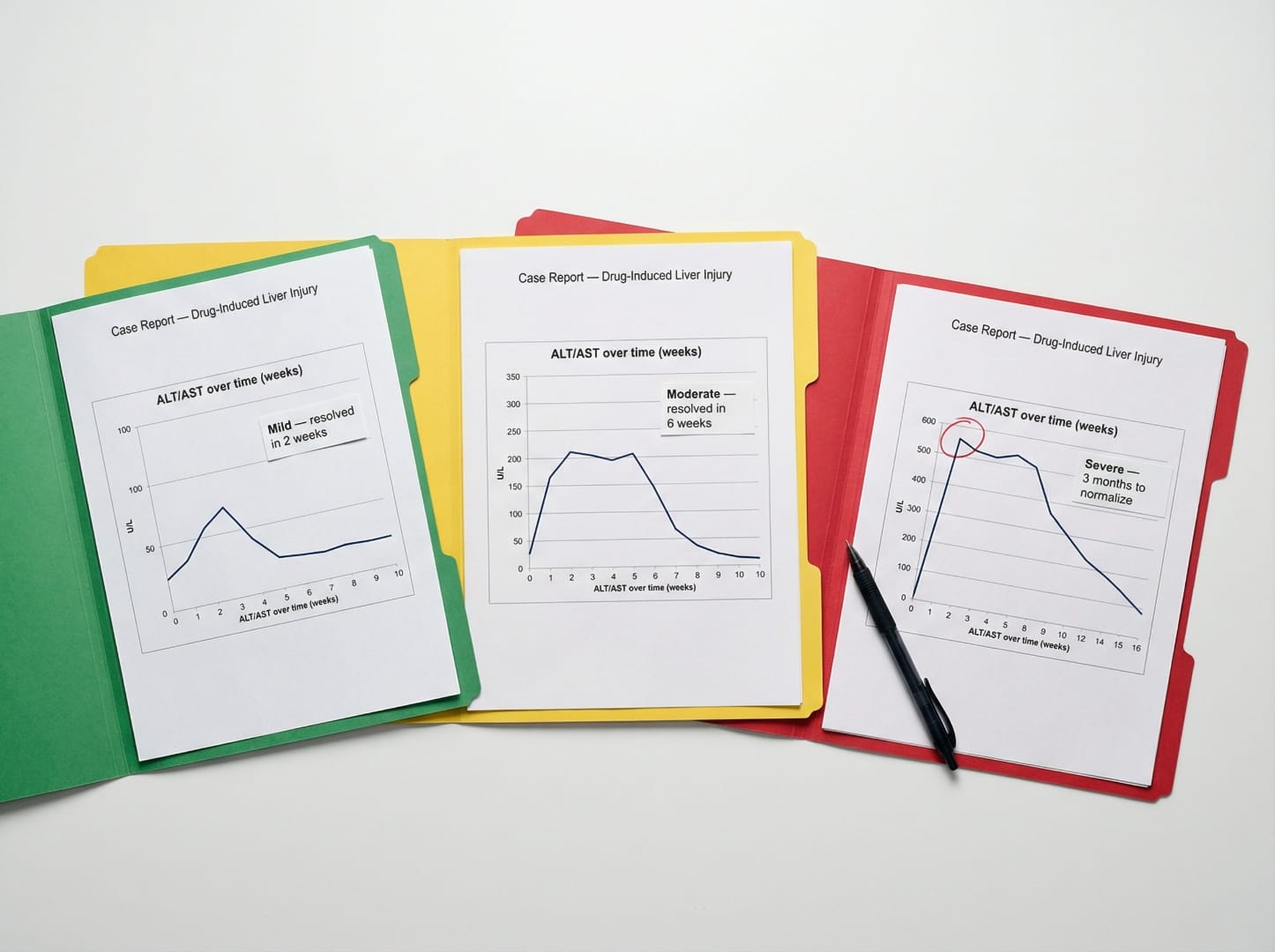
Across all three cases the pattern is consistent: high doses or rapid dose escalation — with or without concurrent ICI — triggered severe but fully reversible hepatocellular injury. No fatalities were reported.
⚠️ Risk Factors: Who Is Most Vulnerable?
Not everyone who takes fenbendazole develops liver injury. The published cases suggest a distinct risk profile:
- ✅ High doses or rapid escalation: All three DILI cases involved FBZ far above the standard 222 mg × 3/week. The standard Joe Tippens dose (666 mg/week) has not been directly associated with DILI in published literature. Rapid escalation — as in Case 1 — appears particularly dangerous.
- ✅ Concurrent ICI therapy: Two of three cases occurred in ICI-treated patients. Heightened immune surveillance amplifies drug-induced hepatic inflammation and complicates attribution.
- ✅ Pre-existing liver conditions: Liver metastases, cirrhosis, active hepatitis, fatty liver, or significant alcohol use reduce hepatic reserve and lower the injury threshold.
- ✅ Concurrent hepatotoxic medications: Methotrexate, azathioprine, isoniazid, antifungals, or other hepatotoxins compete for CYP1A1/1A2 metabolism and increase injury risk.
- ✅ Continuous daily dosing without breaks: Uninterrupted daily use prevents hepatic recovery and glutathione repletion.
- ✅ Absence of monitoring: In Case 2 the patient took FBZ for one year with no labs. Early detection through routine testing is the most effective intervention available.
💡 The 3-On-4-Off Schedule and Liver Safety
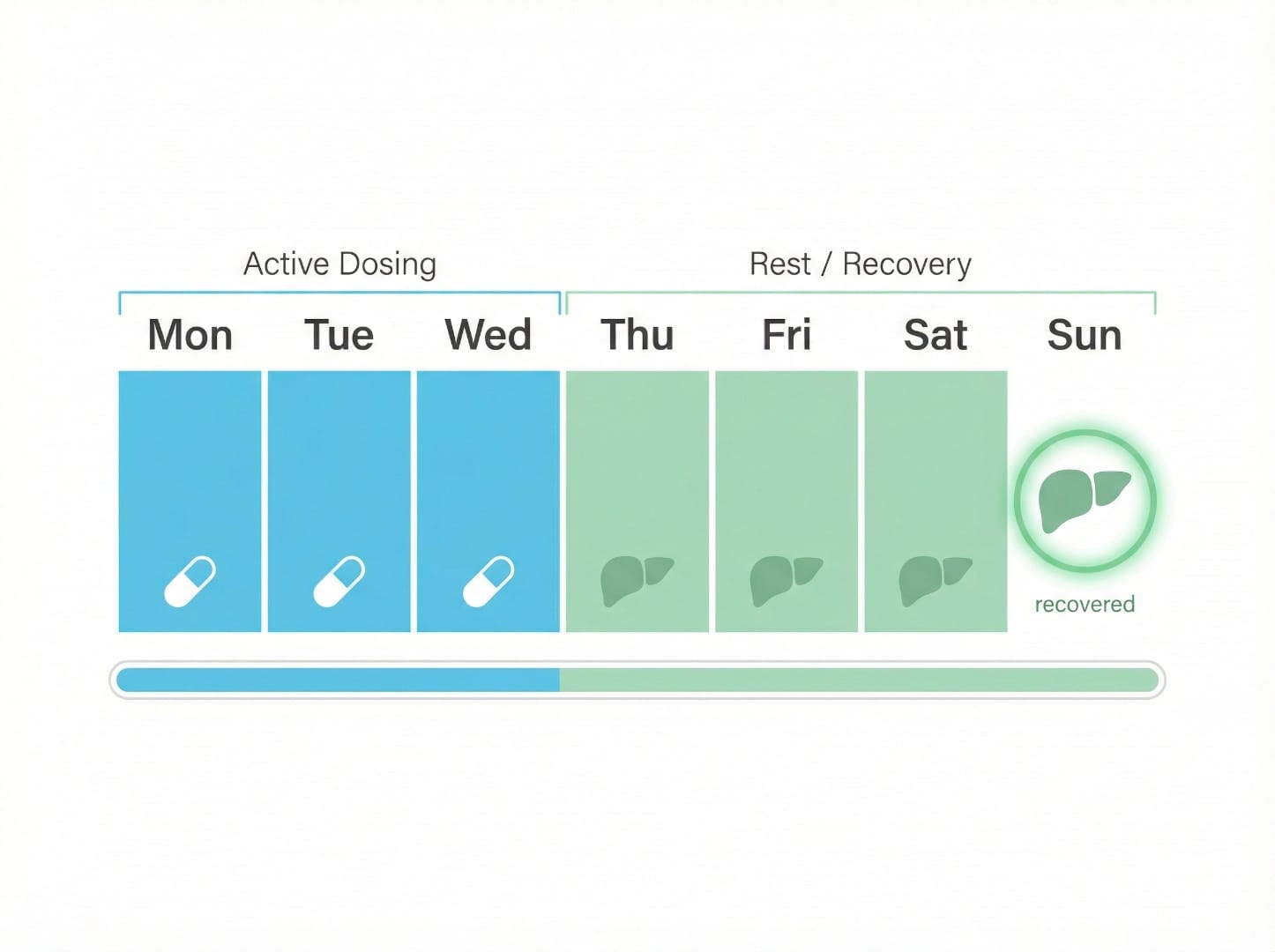
The widely referenced three consecutive days on, four days off schedule provides hepatic recovery time between exposure cycles — allowing glutathione replenishment and metabolite clearance. This is central to the Joe Tippens Protocol, the ISOM Protocol, and the Fenbendazole Dosage Guide. However, Case 3 demonstrates that the schedule alone is not protective at 1 g doses — the key safety formula is standard dose + structured schedule + routine monitoring.
| Protocol Feature | Standard (Low Risk) | High-Risk Pattern |
|---|---|---|
| Dose per day | 222 mg | 1,000 mg+ per day |
| Weekly schedule | 3 days on / 4 days off | Daily (no breaks) |
| Weekly total exposure | 666 mg/week | 1,554–9,000+ mg/week |
| Monitoring | Monthly LFTs for first 3 months | Months with no labs |
| Concurrent ICI | Oncologist-approved, frequent LFTs | Self-initiated without disclosure |
| Liver support | Milk thistle / TUDCA / curcumin | None |
Lab-Tested Fenbendazole from Sanare Lab
99% purity with independent certificates of analysis — quality you can verify before you start.
180 capsules — 99% purity, laboratory tested
180 capsules — higher dose option
120 capsules — with Black Pepper for absorption
Disclaimer: For convenience only. Consult a licensed professional.
🚫 Who Should Avoid Fenbendazole
Clear contraindications and high-risk categories where fenbendazole should be avoided or only used under close medical supervision:
- ⚠️ Severe liver failure or decompensated cirrhosis: Markedly reduced hepatic capacity means even standard doses can accumulate and cause disproportionate injury.
- ⚠️ Active hepatitis (viral or autoimmune): Pre-existing hepatic inflammation dramatically lowers the DILI threshold and may cause rapid decompensation.
- ⚠️ Severe kidney failure: Impaired elimination of metabolites increases systemic exposure to potentially toxic breakdown products.
- ⚠️ Concurrent hepatotoxic drugs: Methotrexate, azathioprine, isoniazid, ketoconazole, or other known hepatotoxins — avoid combining without specialist input.
- ⚠️ Active ICI therapy without oncologist awareness: Two of three DILI cases occurred in ICI-treated patients who self-initiated FBZ. If combining FBZ with ICI therapy, disclose to oncologist and monitor LFTs every 2–4 weeks.
- ⚠️ Significant liver metastases: Reduced functional hepatic reserve lowers the threshold for additional metabolic stress.
- ⚠️ Pregnancy: No human safety data; not recommended.
🌿 Liver Support Supplements
Several natural compounds have established hepatoprotective properties and are commonly used alongside fenbendazole protocols. These complement — but do not replace — routine monitoring.
| Supplement | Primary Mechanism | Typical Dose | Notes |
|---|---|---|---|
| Milk Thistle (Silymarin) | Promotes glutathione synthesis; stabilizes hepatocyte membranes; inhibits lipid peroxidation | 150–250 mg silymarin/day | Most widely referenced in benzimidazole protocols; directly addresses glutathione depletion |
| TUDCA | Stabilizes mitochondrial membranes; reduces ER stress; anti-apoptotic in hepatocytes; improves bile flow | 250–500 mg/day | Used in metabolic oncology protocols; mitochondrial-level hepatoprotection |
| Curcumin (with piperine) | NF-κB inhibition; Nrf2 activation; antioxidant | 360–500 mg/day | Dual rationale: hepatoprotective + potential anticancer synergy; included in ISOM Protocol |
| Alpha-Lipoic Acid (ALA) | Glutathione recycler; broad antioxidant (water and fat-soluble); mitochondrial cofactor | 300–600 mg/day | Directly replenishes glutathione pool; addresses core FBZ hepatotoxicity mechanism |
📋 Liver Monitoring Protocol
The most consistent finding across all three DILI cases is the absence of routine monitoring — in Case 2 the patient took FBZ for a full year without any liver function testing. The following framework is based on clinical principles for monitoring hepatotoxic medications; individual schedules should be established with a physician.
| Timepoint | Tests | Action Threshold |
|---|---|---|
| Baseline | Full CMP: AST, ALT, ALP, GGT, total bilirubin, albumin, creatinine | ALT/AST >2× ULN: discuss with physician before initiating |
| Week 2–4 | AST, ALT, ALP, total bilirubin | Priority on ICI therapy or any baseline liver abnormality |
| Month 1 | Full CMP | AST/ALT >3× ULN: pause FBZ, recheck in 1–2 weeks; still elevated → stop and consult |
| Month 2 | AST, ALT, ALP, bilirubin | AST/ALT >5× ULN anytime: stop immediately, urgent medical evaluation |
| Month 3 | Full CMP | Stable normal LFTs for 3 months: transition to quarterly |
| Quarterly (month 4+) | AST, ALT, ALP, bilirubin, creatinine | Return to monthly if dose is escalated |
| Anytime (urgent) | Immediate if: jaundice, dark urine, pale stools, severe fatigue, right upper quadrant pain, unexplained nausea | STOP fenbendazole immediately — do not wait for scheduled testing |
Patients on ICI therapy should discuss monitoring frequency with their oncologist before starting FBZ. For additional guidance on dosing schedules, see the Fenbendazole Dosage Guide and real-world user accounts at Customer Notes & Experiences.
✅ Putting It in Perspective
Three peer-reviewed case reports document serious FBZ-associated DILI — but it is equally important to acknowledge what the evidence does not show. All three cases involved doses substantially above the standard 222 mg × 3/week regimen, rapid escalation, or concurrent high-risk conditions. In all three cases, liver injury was fully reversible — normalization occurred within 7 weeks to 3 months after stopping. No permanent damage, no fatalities attributable to FBZ hepatotoxicity. Thousands of patients have reported using FBZ at standard doses without liver complications (see Customer Notes & Experiences). The combination of standard dosing, 3-on-4-off scheduling, liver-supportive supplements, and routine LFT monitoring addresses every identified risk factor in the published literature.
🔗 Key Takeaways
- Three peer-reviewed case reports (2021, 2024, 2026) document fenbendazole-induced hepatocellular DILI, with RUCAM/Naranjo scores of 6–9.
- All cases involved doses substantially above the standard 222 mg × 3/week protocol — Case 1's trigger was a 2.3× increase in weekly exposure by switching to daily use.
- Liver injury was fully reversible in all three cases — normalization within 7 weeks to 3 months after stopping FBZ.
- Primary risk factors: high doses, rapid escalation, concurrent ICI therapy, pre-existing liver disease, and no routine monitoring.
- The 3-on-4-off schedule reduces risk by allowing hepatic recovery between cycles — but does not protect against injury at supra-standard doses.
- Liver-supportive supplements — milk thistle, TUDCA, curcumin, alpha-lipoic acid — are reasonable and evidence-supported additions to any sustained FBZ protocol.
- Baseline LFTs before starting, then monthly for 3 months, then quarterly, is the minimum appropriate monitoring framework.
- Contraindications: severe liver/kidney failure, active hepatitis, concurrent hepatotoxic drugs — avoid or use only under close specialist supervision.
- Most people at standard doses (222 mg, 3×/week) do not develop liver problems. The published cases are important safety signals, not evidence of widespread toxicity at recommended doses.
Disclaimer: This article is for educational and research purposes only. It does not constitute medical advice. Fenbendazole is not FDA-approved for human use. Always discuss any investigational protocol with a qualified physician, particularly if you are on immunotherapy or have pre-existing liver or kidney conditions.
Scientific References
- Krishnan A, Lucas K, Maas L, Woreta TA. (2026). Differentiating fenbendazole-induced liver injury from immune-mediated hepatitis in a patient with metastatic colon cancer on nivolumab/relatlimab. World J Clin Cases. PMC12836008
- Thakurdesai A, Rivera-Matos L, Nagra N, Busch B, Mais DD, Cave MC. (2024). Severe Drug-Induced Liver Injury Due to Self-administration of the Anthelmintic Fenbendazole. ACG Case Rep J. PMC11068125
- Yamaguchi T, Shimizu J, Oya Y, Horio Y, Hida T. (2021). Drug-Induced Liver Injury in a Patient with Nonsmall Cell Lung Cancer after the Self-Administration of Fenbendazole Based on Social Media Information. Case Rep Oncol, 14(1), 886–891. PMC8255718
- Fenbendazole.org. Fenbendazole Safety and Side Effects. fenbendazole.org
- Dogra N, Kumar A, Mukhopadhyay T. (2018). Fenbendazole acts as a moderate microtubule destabilizing agent and causes cancer cell death. Sci Rep (Nature), 8, 11926. PMC6098001
Shop Sanare Lab
Below are commonly referenced items. Links are provided for convenience — always review the label and consult a professional before use.
180 capsules — 99% purity, laboratory tested
180 capsules — higher dose option
120 capsules — with Black Pepper for absorption
6 / 12 / 18 mg — 100 tablets
Disclaimer: Links are informational and for convenience. This site does not provide medical advice and does not endorse any specific vendor. Always verify product quality, labeling, and consult a licensed professional for health decisions.