In May 2025, a peer-reviewed case series published in Case Reports in Oncology documented something that oncologists and cancer researchers rarely see: three patients with Stage IV cancers — breast, prostate, and melanoma — who achieved complete or near-complete remission while self-administering fenbendazole, an inexpensive veterinary antiparasitic. The paper, authored by Dr. William Makis, Ilyes Baghli, and Pierrick Martinez, presented detailed biomarker data, serial imaging, and multi-year follow-up. The data itself was striking. Then, in January 2026, Karger retracted the paper — not because the clinical data was incorrect, but because the lead author had failed to disclose a financial conflict of interest.
This article is a careful, detailed walkthrough of what those three cases actually showed — alongside a fourth case of Stage IV diffuse large B-cell lymphoma (DLBCL) that regressed on fenbendazole monotherapy — and what the data can and cannot tell us. We examine the retraction controversy, the skeptics' most credible arguments (including Dr. Roman's pointed YouTube analysis), and what the scientific community says would be required to move fenbendazole from compelling anecdote to evidence-based oncology. For context on the broader fenbendazole landscape, see our Joe Tippens Protocol overview and our Fenbendazole Dosage Guide.
⚠️ Educational Disclaimer
This article is for research and informational purposes only. It does not constitute medical advice and is not a substitute for professional oncology consultation. Always discuss any investigational protocol with your physician.
📋 About the 2025 Case Series (PMC12215191)
The paper — titled "Fenbendazole as an Anticancer Agent? A Case Series of Self-Administration in Three Patients" — was received by Case Reports in Oncology on January 31, 2025, accepted May 7, 2025, and published online May 26, 2025 (PMC12215191). It carried a PMID of 40605964 and was assigned DOI 10.1159/000546362. The authors noted in their discussion that this was "only the second case series documenting such outcomes," following an earlier Stanford University Medical Center series led by Chiang et al.
The paper followed the CARE Checklist for case reports and included online supplementary material with full medical records for each patient. The authors were affiliated with Alberta Health Services / Cross Cancer Institute (Edmonton, Canada), the International Society for Orthomolecular Medicine (Toronto, Canada), and the Association Cancer et Métabolisme (Nîmes, France). The corresponding author was Pierrick Martinez.
Key design points readers should understand upfront:
- All three patients self-administered FBZ — this was not a controlled trial or physician-directed therapy
- All three patients used FBZ alongside other treatments (hormonal therapy, ADT, immunotherapy, radiation, or surgery)
- The case series was retrospective and descriptive — it observed outcomes, not controlled for confounders
- The authors explicitly state the data is "hypothesis-generating only" and does not establish causation
- The paper was retracted in January 2026 due to undisclosed conflict of interest by the lead author — the clinical data itself was not disputed
With those caveats firmly in place, here is what the data showed.
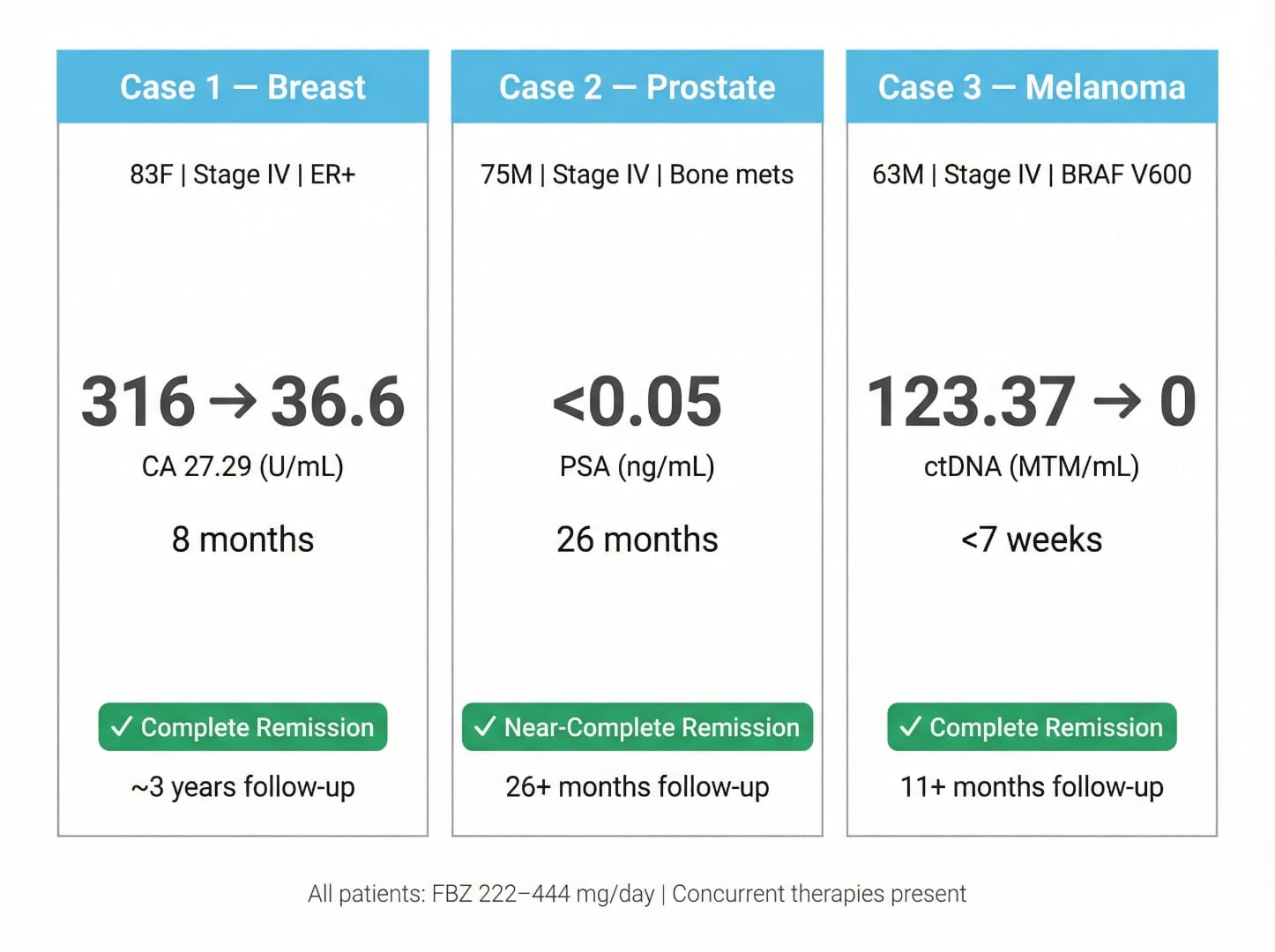
🩺 Case 1: Stage IV Breast Cancer — Complete Remission at 3 Years
The first patient was an 83-year-old woman with a long oncological history. She had originally been diagnosed with estrogen receptor-positive (ER+) breast cancer in 2009 and underwent bilateral mastectomy, reconstruction, and aromatase inhibitor therapy. She remained disease-free for over a decade before a recurrence was confirmed in October 2021.
Her 2021 recurrence represented advanced, multisystem metastatic disease. Imaging revealed metastatic involvement of the liver, lungs, and bones. MRI of the spine showed metastatic breast cancer at T10, T12, L1, L2, L3, L4, L5, S1, S2, and the iliac bones. Ascitic fluid analysis confirmed metastatic breast carcinoma. A PET/CT on December 29, 2021 revealed six hypermetabolic lung lesions (largest: 2.8 × 1.5 cm, SUV max 8.4), plus additional lesions in the right lower lobe and left upper lobe. Immunohistochemistry confirmed ER/PR positive, HER-2/neu negative. CA 27.29 (breast cancer tumor marker) was 316 in November 2021 — well above the normal ceiling of 38 U/mL.
The patient declined conventional chemotherapy and was placed under hospice care. On November 22, 2021, she began self-administering FBZ at 222 mg/day. In December 2021, she received fulvestrant injections (an estrogen receptor blocker). In January 2022, she underwent targeted radiation for two painful spinal metastases — the tumors disappeared rapidly, relieving her pain within days. She also continued supplementation with vitamin D 5,000 IU and a multivitamin throughout.
The biomarker and imaging response was dramatic:
| Date | Event / Measurement | Result |
|---|---|---|
| October 2021 | Recurrence confirmed; MRI spine | Multi-bone metastases |
| November 22, 2021 | FBZ 222 mg/day started; CA 27.29 | CA 27.29 = 316 U/mL |
| December 2021 | Fulvestrant injections begin; PET/CT | Hypermetabolic lung/liver/bone lesions |
| January 2022 | Targeted radiation (2 spinal mets) | Pain relief within days |
| April 20, 2022 | PET scan | No abnormal metabolic activity — NED |
| July 2022 | CA 27.29; liver enzymes | CA 27.29 = 36.6 U/mL (normal); transient ALT/AST elevation (normalized within weeks) |
| June 2022 | All treatment discontinued | Confirmed complete remission (NED) |
| February 2023 | CA 27.29 follow-up | CA 27.29 = 26.5 U/mL (normal) |
| ~2024 (ongoing) | Follow-up monitoring; continues FBZ | Remains recurrence-free, ~3 years post-remission |
The CA 27.29 drop from 316 → 36.6 over 8 months — crossing the normal threshold — coincided with complete metabolic disappearance of tumors on PET imaging. The authors noted that CA 27.29 can lag cancer elimination by several months, and that the transient liver enzyme elevation in July 2022 (after remission was already confirmed) may have been attributable to fulvestrant, FBZ, or an interaction between the two. No adverse effects were directly attributable to FBZ at 222 mg/day during the treatment period. The patient remains recurrence-free and continues taking FBZ daily nearly 3 years after being declared in complete remission.
🩺 Case 2: Stage IV Prostate Cancer — Near-Complete Remission at 26 Months
The second patient was a 75-year-old man with recurrent Stage IV prostate cancer and extensive bone metastases. He had originally been diagnosed approximately 10 years earlier (around 2011) and treated surgically. He maintained undetectable PSA levels for 18 months before a gradual PSA rise indicated recurrence. By December 2021, the extent of disease was confirmed through imaging: bone metastases in the spine, pelvic bones, and right humeral head, along with lymph node involvement. A December 16, 2021 CT scan identified prominent left periaortic lymph nodes (representative node: 0.8 cm) not previously seen.
In December 2021, the treating team initiated androgen deprivation therapy (ADT) with Orgovyx (relugolix) and Erleada (apalutamide), complemented by Xgeva (denosumab) for bone health. The patient also added a comprehensive supplement regimen: vitamin D 5,000–10,000 IU/day with K2 and magnesium, melatonin 10–40 mg/day, berberine, curcumin, artemisinin, cimetidine, and other compounds with putative anticancer effects. He began self-administering FBZ in December 2021 at a dose range of 222–444 mg/day, usually daily with occasional dose reductions.
| Date | Event / Measurement | Result |
|---|---|---|
| December 2021 | Recurrence confirmed; CT scan; ADT + FBZ started | Bone mets (spine, pelvis, humerus); left periaortic LN 0.8 cm |
| December 2022 (12 months) | CT follow-up | Bone lesion regression; lymph nodes fully resolved |
| January 2024 (24 months) | CT follow-up | Significant bone lesion regression; no new metastatic sites |
| April 2024 (28 months) | PSMA-PET/CT whole-body scan | Vast majority of sclerotic bone lesions: no abnormal radiopharmaceutical uptake; no LN uptake |
| 26+ months (ongoing) | PSA monitoring; continues FBZ + ADT/Xgeva | PSA undetectable <0.05 ng/mL for >2 years; near-complete response sustained |
The April 2024 PSMA-PET/CT was the most definitive imaging result: the vast majority of previously active bone lesions showed no abnormal radiopharmaceutical accumulation, consistent with near-complete metabolic remission. A large left renal cortical cyst showed SUV of 0.5. PSA remained undetectable for over 26 months. The authors reported no increase in liver enzymes or any other side effects attributable to FBZ throughout the treatment period. The patient remains in near-complete response and continues both FBZ and ADT/Xgeva.
⚠️ Critical caveat: As discussed below, this case presents the most significant interpretive challenge. The ADT regimen used — specifically Erleada (apalutamide) — is one of the most potent agents in metastatic prostate cancer, and undetectable PSA is an expected, not surprising, outcome in many patients on this combination.
🩺 Case 3: Stage IV Melanoma — Complete Remission via ctDNA
The third patient was a 63-year-old man with BRAF V600-mutated melanoma. He was initially diagnosed in July 2020 with Stage IIIC disease (hip growth) and began 8 months of adjuvant dabrafenib and trametinib, stopping early in May 2021 due to decreased ejection fraction. He achieved remission, which lasted until 2023.
On December 12, 2023, a biopsy confirmed recurrence: a 1.6 mm ulcerated malignant melanoma in the lower left abdomen (SOX-10 and pan-melanoma positive). PET-CT revealed multiple hypermetabolic foci — peritoneal and retroperitoneal nodules, focal uptake in the stomach and small bowel, lesions in the right gluteus medius, quadratus femoris, and L5 vertebra. An incidental finding revealed mass-like distal ureter thickening, subsequently biopsied and confirmed as a separate malignancy (urothelial carcinoma). Tempus xF circulating tumor DNA (ctDNA) testing confirmed the presence of BRAFV600 mutation, consistent with recurrent melanoma.
The patient's oncologist recommended delaying immunotherapy with nivolumab (Opdivo) pending further biopsy. This created a treatment-free window, during which the patient began self-administering FBZ in mid-December 2023. Surgery in mid-December 2023 addressed the ureteral tumors disrupting urination. The patient subsequently received two doses of nivolumab and supplements including ascorbic acid 2g BID, cephalexin, cholecalciferol, CoQ10, cyanocobalamin, and glutathione.
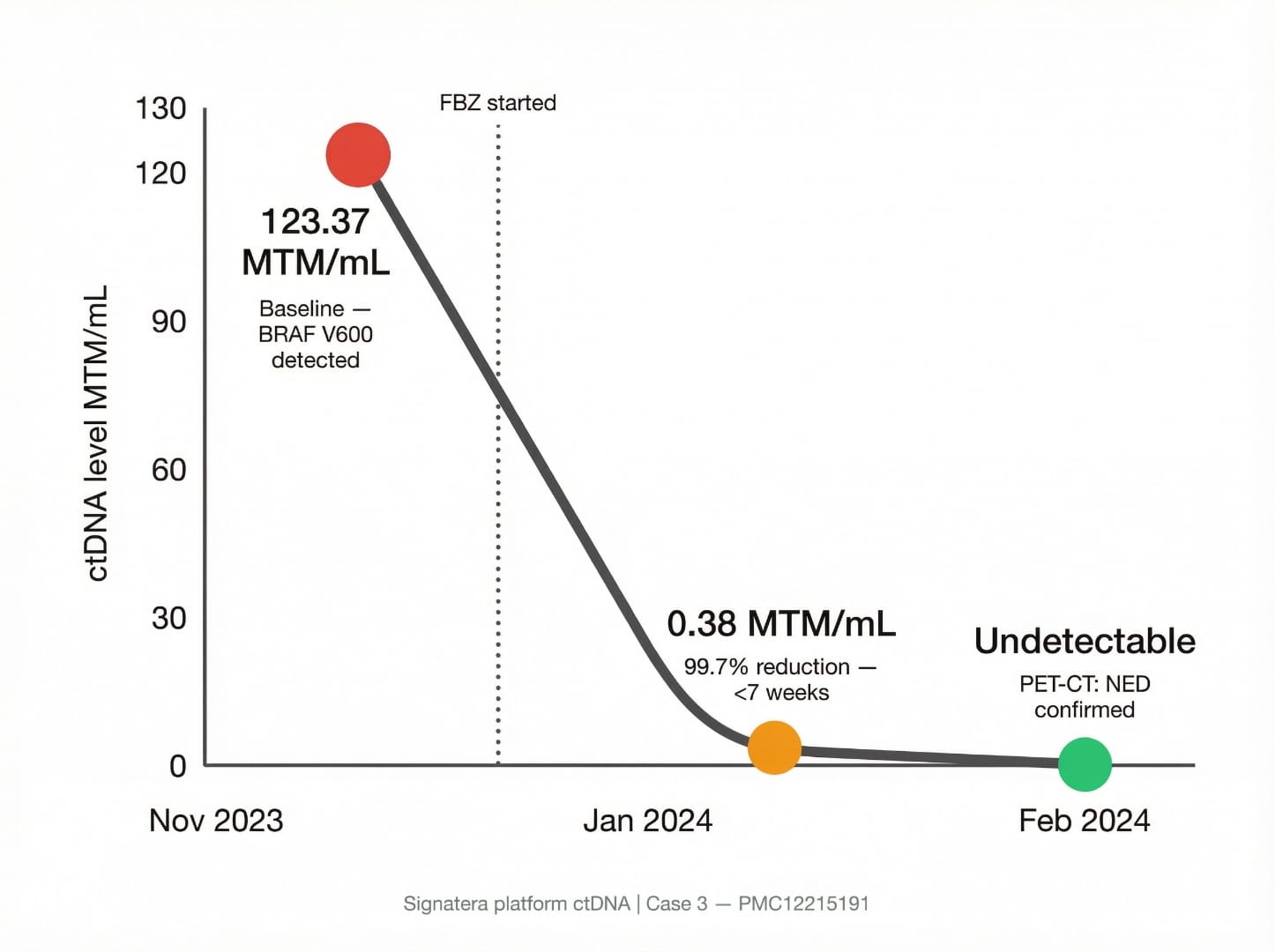
The ctDNA data (Signatera platform) provided unusually precise quantification of the response:
| Date | Event | ctDNA / Signatera Result |
|---|---|---|
| November 29, 2023 | Baseline — before FBZ start | 123.37 MTM/mL (BRAFV600 detected) |
| Mid-December 2023 | FBZ 222 mg/day started; ureteral surgery; FBZ dose escalated to 444 mg/day during this period | — |
| January 17, 2024 (<7 weeks post-FBZ) | ctDNA follow-up | 0.38 MTM/mL — 99.7% reduction |
| February 21, 2024 | ctDNA follow-up; PET-CT | 0 (undetectable); PET-CT: NED (no evidence of disease) |
| November 2024 (11+ months) | Follow-up | Remains melanoma recurrence-free |
The drop from 123.37 → 0.38 in under 7 weeks, followed by complete ctDNA clearance and NED on PET-CT, was among the fastest biomarker responses in any published fenbendazole case report. The authors noted that this occurred during what was effectively a treatment-free window from standard melanoma therapy (no active BRAF inhibitor, and only two doses of nivolumab were administered during the response period, the first of which came after the dramatic initial ctDNA drop). The patient has remained melanoma recurrence-free for 11+ months.
📊 Summary Comparison: All Three Cases at a Glance
The table below consolidates the key parameters across all three cases to facilitate side-by-side comparison:
| Parameter | Case 1 — Breast | Case 2 — Prostate | Case 3 — Melanoma |
|---|---|---|---|
| Patient | 83-year-old female | 75-year-old male | 63-year-old male |
| Diagnosis | Stage IV ER+ breast cancer; mets to liver, lungs, bones, ascites | Stage IV prostate cancer; bone mets (spine, pelvis, humerus); lymph node involvement | Stage IV BRAF V600-mutated melanoma; peritoneal, retroperitoneal, GI, spinal mets |
| Prior treatment | Bilateral mastectomy (2009); aromatase inhibitors; declined chemo | Prostatectomy (~2011); PSA remission 18 months then recurred | Dabrafenib/trametinib (2020–2021, stopped early); pembrolizumab (prior cycle) |
| FBZ dose | 222 mg/day | 222–444 mg/day | 222–444 mg/day |
| FBZ start | November 22, 2021 | December 2021 | Mid-December 2023 |
| Concurrent therapies | Fulvestrant (Dec 2021); targeted radiation x2 spinal mets (Jan 2022); Vitamin D 5,000 IU; multivitamin | ADT: Orgovyx + Erleada; Xgeva; Vitamin D, K2/Mg, melatonin, berberine, curcumin, artemisinin, cimetidine | Ureteral surgery (Dec 2023); 2 doses nivolumab; ascorbic acid 2g BID, CoQ10, glutathione, cholecalciferol |
| Key biomarker change | CA 27.29: 316 → 36.6 (8 months) | PSA: undetectable (<0.05 ng/mL) for 26+ months | ctDNA (Signatera): 123.37 → 0.38 in <7 weeks → 0 by Feb 2024 |
| Imaging outcome | PET Apr 2022: no abnormal metabolic activity; subsequent PETs NED | CT Dec 2022: bone regression, LN resolved; PSMA-PET Apr 2024: majority of lesions no uptake | PET-CT Feb 2024: NED |
| Outcome | ✅ Complete remission | ✅ Near-complete remission | ✅ Complete remission |
| Follow-up duration | ~3 years (continues FBZ) | 26 months (continues FBZ + ADT) | 11+ months (continues FBZ) |
| FBZ adverse effects | None reported; transient liver enzyme elevation (cause unclear) | None reported | None reported |
🔬 The DLBCL Case: FBZ as Monotherapy
The PMC12215191 discussion section references a fourth case that is arguably more provocative from a mechanistic standpoint: a published case report of Stage IVa diffuse large B-cell lymphoma (DLBCL) in which the patient declined chemotherapy entirely and achieved disease regression on fenbendazole monotherapy at 1 g/day.
In this case, an 83-year-old male presented with heartburn, difficulty swallowing, and fatigue. Esophagogastroduodenoscopy revealed a superficial duodenal ulcer that biopsied as DLBCL (GCB subtype; FISH negative for c-MYC, BCL2, BCL6). A PET/CT staged him at IVa, showing hypermetabolic activity in the gastric antrum/proximal duodenum, periaortic lymph node, and pulmonary nodules. The patient was offered chemotherapy but declined due to concerns about side effects, and instead began self-researching and initiated FBZ at approximately 1 g/day.
Key timeline of the DLBCL case:
- After 6 months on 1–6 tabs/day of FBZ, he tapered to 1–3 tabs daily due to peripheral neuropathy (a notable adverse effect not seen in the Makis series)
- Repeat CT revealed smaller mediastinal lymph nodes
- Subsequent PET/CT showed improved lymphadenopathy from prior scans
- A further PET/CT 3 months later (patient now at 3 tabs weekly) revealed interval improvement with no new lesions
- After 12 months of follow-up, continued disease regression with no chemotherapy administered
The significance of this case is that it removes the confounding variable of concurrent conventional therapy. In all three cases in PMC12215191, other therapies were present. In the DLBCL case, FBZ operated largely as a solo agent — though the authors of that report still acknowledge that whether the regression was due to FBZ or other factors "still unknown." The case was published by Abughanimeh, Evans, and Kallam at the University of Nebraska Medical Center / Fred & Pamela Buffett Cancer Center (Austin Publishing Group, 2020). DLBCL, notably, can in rare cases undergo spontaneous remission — a significant caveat.
For readers interested in how fenbendazole compares mechanistically with other antiparasitic agents investigated in oncology, our article on Ivermectin in Cancer Protocols provides a useful parallel reference.
Fenbendazole from Sanare Lab
The same 222 mg and 444 mg dosages used in the case reports above — pharmaceutical-grade, lab-tested at 99% purity with certificates of analysis.
180 capsules — 99% purity, laboratory tested
180 capsules — higher dose option
Disclaimer: For convenience only. Consult a licensed professional before use.
⚠️ Limitations and Controversy: What the Critics Say
The case series attracted significant critical commentary — some measured and scientifically grounded, some more polemical. The most substantive critique came from a March 2026 YouTube review by Dr. Roman, a urologist, who dissected each case systematically (Fenbendazole and Cancer: Evidence, Myths & Risks!). His critique deserves detailed engagement because it raises legitimate methodological concerns.
On Case 2 (Prostate Cancer): Dr. Roman's sharpest point was directed at the prostate cancer case. He argued that an undetectable PSA in a patient on Orgovyx (relugolix) and Erleada (apalutamide) is not a surprising finding — it is an expected one. In the original TITAN trial for apalutamide in metastatic castrate-sensitive prostate cancer, 68% of patients achieved undetectable PSA compared to 28% with standard ADT alone. "That has nothing to do with the fenbendazole," Dr. Roman stated. "That's a normal response from these drugs." He described the authors' framing that "the use of fenbendazole coincided with continued regression of metastatic lesions" as "outrageous" given the known efficacy of the ADT combination.
On Case 3 (Melanoma): Dr. Roman highlighted that the melanoma patient received two doses of nivolumab — a PD-1 inhibitor (Opdivo) — during the response period. He noted that nivolumab is "an incredibly potent immunotherapy agent" that has transformed 5-year melanoma survival from <5% to over 50%. "To see a response from this drug is not unsurprising," he argued, questioning whether FBZ played any role at all.
On Case 1 (Breast Cancer): Dr. Roman did not address the breast cancer case in comparable depth in the YouTube review, though fulvestrant — the estrogen receptor antagonist used — is itself an established therapy for ER+ metastatic breast cancer, and targeted radiation to painful spinal metastases was also administered.
Beyond Dr. Roman's critique, the scientific limitations the authors themselves acknowledged include:
- 📌 Small sample size: Three cases cannot generate statistical inference
- 📌 No control group: No comparator patients receiving identical conventional therapies without FBZ
- 📌 Retrospective design: Data was collected after outcomes were already known, introducing selection bias
- 📌 Self-selection: Patients who self-administer alternative compounds may differ systematically from those who do not (health motivation, compliance, supplement use, etc.)
- 📌 Confounding therapies: The impossibility of isolating FBZ's contribution from that of concurrent conventional or supplementary treatments is the central methodological problem
- 📌 Spontaneous remission: While rare, spontaneous remission is documented in all three cancer types (breast, prostate, melanoma), and cannot be excluded
- 📌 Unknown pharmacokinetics in these patients: No plasma FBZ levels were measured; actual drug exposure is unknown
📰 The Retraction Controversy: What Karger Said — and What It Didn't
On January 21, 2026, Karger Publishers issued a formal retraction notice (Case Rep Oncol. 2026;19(1):169; also indexed at PMC12823103).
The critical point: the retraction was not based on disputed or falsified clinical data. According to reporting by Marchward, the stated reason was undisclosed conflict of interest on the part of lead author William Makis. Specifically, Makis had been offering coaching services and selling fenbendazole products on a website operated in his name during the period when the case study was submitted and published — a direct financial conflict of interest that was not declared in the paper.
Makis responded publicly, framing the retraction as "Big Pharma attacking" the paper. Marchward noted that if the conflict of interest allegation is accurate, the retraction is "entirely valid" under scientific publishing standards — while also acknowledging that such non-disclosure is regrettably common in the broader scientific literature, and that it is "not unreasonable that 'Big Pharma' has leveraged this improper scientific conduct to effectuate a retraction."

What this means for interpreting the data:
- ✅ The clinical records and biomarker data were not challenged as inaccurate or fabricated
- ✅ The three patients and their documented outcomes remain on the medical record
- ⚠️ The conflict of interest introduces a legitimate question about whether the cases were selected or framed to support a commercial narrative
- ⚠️ Retracted papers cannot be cited in subsequent academic work as if they were current peer-reviewed evidence
- 📌 The PMC entry for PMC12215191 remains accessible but is flagged: "This article has been retracted"
The paper continues to be covered by medical journalism outlets including Medindia, which reported on the case series as a scientifically significant signal warranting further investigation. The data did not disappear with the retraction; the cases happened. What the retraction changes is the epistemic weight those observations can carry in formal scientific argumentation.
📈 The Broader Picture: How Many Cancer Patients Are Using FBZ?
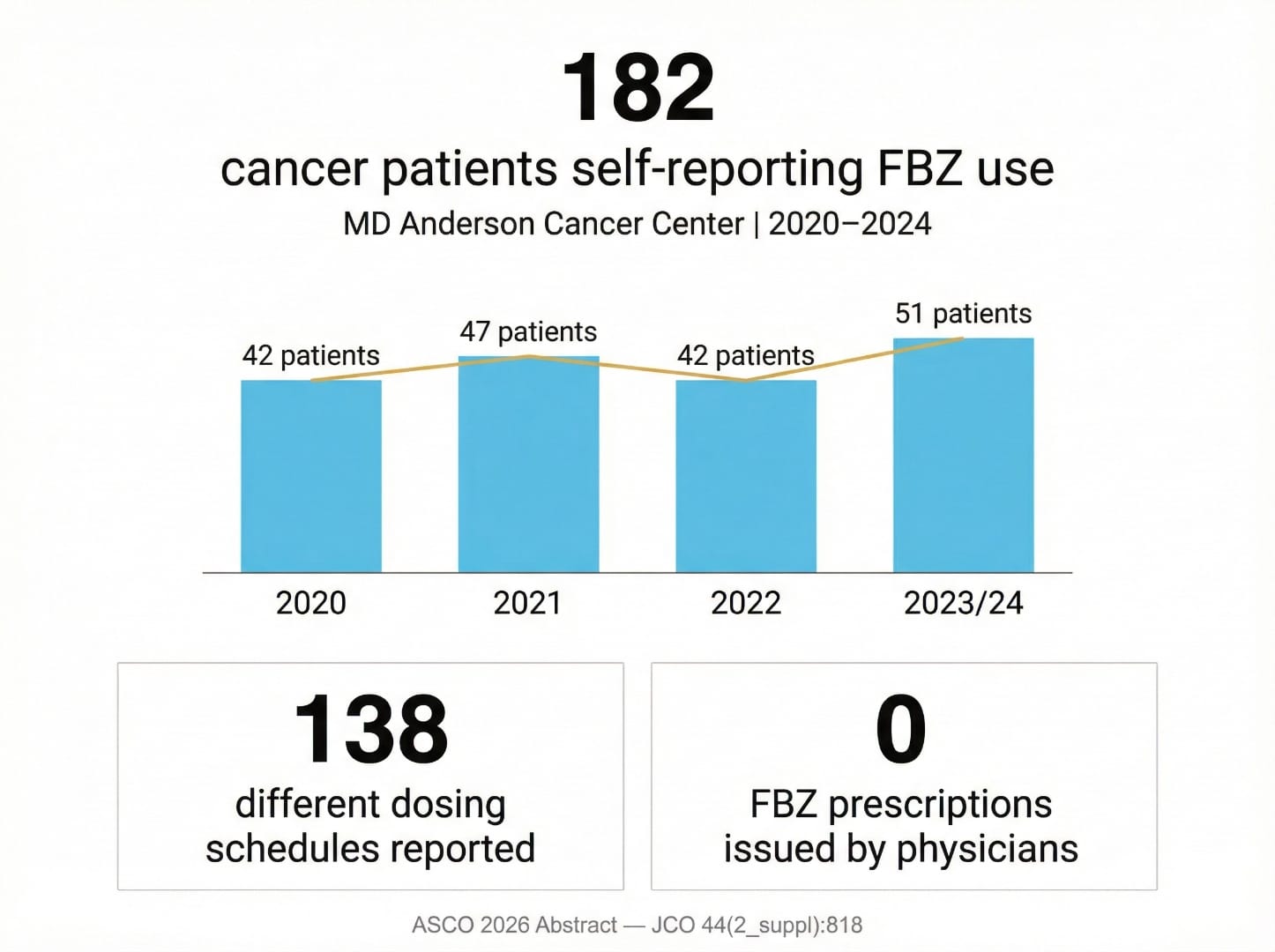
The case series exists within a larger context of patient-led fenbendazole use that has expanded significantly since the Joe Tippens story went viral in 2019. A 2026 ASCO abstract by Phan, Willis, and Hu from MD Anderson Cancer Center (MDACC) provides the most rigorous institutional data yet on real-world FBZ use patterns (JCO 2026;44(2_suppl):818).
The MDACC retrospective analysis covered 297,223 patients visiting the institution from January 2020 to January 2024 who self-reported non-prescription medication use. Key findings:
- 182 patients (0.06% of all MDACC visitors) self-reported taking fenbendazole as supplemental therapy
- Annual FBZ use at MDACC: 42 (2020), 47 (2021), 42 (2022), 51 (2023/partial 2024) — a steady trend
- Most common cancers among FBZ users: metastatic disease, prostate cancer, colorectal cancer
- Among GI malignancies specifically: colorectal, pancreatic, and liver metastases were most represented
- 138 different dosages and frequencies were reported — illustrating the absence of any standardized clinical protocol
- No FBZ prescriptions were issued by MDACC physicians — all use was patient-initiated
- 499 patients self-reported ivermectin use in the same period (higher, partly attributable to COVID-era interest)
The MDACC data is important for understanding the practical context. These patients are using FBZ at one of the world's top cancer centers, alongside their conventional treatments, without telling their physicians in most cases. The wide dosage variation (138 different reported schedules) underscores the absence of evidence-based dosing guidance. For a deeper look at the dosing evidence base, our Fenbendazole Dosage Guide compiles the available protocol references.
Separately, prior surveys of social-media-using cancer patients have suggested that 15–20% have used or seriously considered fenbendazole — a striking figure that reflects the compound's online reach well beyond formal healthcare channels.
⚗️ What the Science Would Need to Establish FBZ as a Cancer Therapy
Both the authors of PMC12215191 and external commentators including the Medindia analysis agree on what would be required for fenbendazole to move from compelling anecdote to evidence-based oncology. The pathway is well-established:
- Pharmacokinetic studies in cancer patients: FBZ's bioavailability in humans is poorly characterized. Studies would need to establish how the drug is absorbed, what plasma concentrations are achieved, how it is metabolized (likely via CYP enzymes), and how it is cleared. Animal data suggests variable oral bioavailability that may differ substantially from the doses used in case reports.
- Phase I dose-escalation trials: Before efficacy can be evaluated, safe dose ranges in cancer patients must be established. A mebendazole Phase I trial (the closest structural analogue) found that 4 of 15 patients taking 200 mg/day experienced liver damage requiring dose reduction — suggesting hepatotoxicity must be carefully monitored in formal trials.
- Biomarker identification: Which patients are most likely to respond? Which cancer types express the molecular targets (tubulin isoforms, GLUT transporters, p53 pathways) that FBZ is hypothesized to act on? Identifying predictive biomarkers is essential for designing trials with adequate power and for avoiding unnecessary exposure in patients unlikely to benefit.
- Drug-drug interaction assessment: Case 2 patients were receiving complex multi-drug regimens. Pharmacokinetic interactions between FBZ and apalutamide, relugolix, or denosumab have not been characterized. Interaction studies are mandatory before combination trials can proceed.
- Randomized controlled trials with FBZ as the experimental arm: Ultimately, only a randomized design with matched control arms receiving identical conventional therapy without FBZ can establish whether FBZ contributes independently to outcomes.
For comparison, see how similar evidence questions have been investigated in the ivermectin oncology space, including the Phase I/II clinical trial NCT05318469 at Cedars-Sinai Medical Center, which represents one of the few formal human oncology trials for an antiparasitic repurposed agent. Fenbendazole is not yet at that stage.
🔗 Connecting the Evidence: What Case Reports Can and Cannot Tell Us
Medical journalism sometimes treats case reports as either definitive proof or meaningless noise. The reality is more nuanced. Case reports occupy a specific and legitimate role in the evidence hierarchy: they are hypothesis-generating signals, not proof of efficacy. The history of oncology includes multiple examples of drugs first observed in case reports that went on to become standard therapy — and far more examples of case report signals that failed to replicate in controlled trials.
What the four fenbendazole cases above — the PMC12215191 series and the DLBCL case — collectively demonstrate:
- ✅ FBZ is tolerated by most patients at 222–444 mg/day without severe acute toxicity
- ✅ FBZ can be administered concurrently with hormonal therapy, ADT, and immunotherapy without obvious pharmacological catastrophe in these four cases
- ✅ Dramatic clinical responses — complete NED on PET imaging, ctDNA clearance to zero — were documented in patients who used FBZ
- ⚠️ FBZ cannot be identified as the causative agent in any of the four cases; co-treatments with known efficacy are present in each
- ⚠️ The dataset is too small and uncontrolled to generate any meaningful estimate of response frequency
- ⚠️ Selection bias is substantial — only patients with dramatic outcomes become case reports
It is also worth noting the counterweight to the optimistic narrative: Scott Adams, the creator of the Dilbert comic strip, publicly discussed taking fenbendazole for his prostate cancer — and subsequently reported that his cancer continued to progress. Cases like Adams's rarely make it into peer-reviewed literature precisely because they are not dramatic positive outcomes. The publication bias in case reports systematically overrepresents favorable responses.
For readers considering the full landscape of repurposed antiparasitic agents in cancer, our comparison of Fenbendazole vs. Mebendazole provides mechanistic context, and our Customer Notes & Experiences page documents firsthand accounts from individuals using these compounds.
Key Takeaways
- 📌 The PMC12215191 case series (Makis, Baghli, Martinez, 2025) documented complete or near-complete remission in three Stage IV cancer patients (breast, prostate, melanoma) who self-administered fenbendazole at 222–444 mg/day alongside conventional therapies
- 📌 A fourth published case (DLBCL, University of Nebraska, 2020) documented disease regression in a patient who declined chemotherapy and used fenbendazole at 1 g/day as monotherapy for 12+ months
- 📌 The case series was retracted by Karger in January 2026 due to undisclosed conflict of interest by the lead author — the clinical data itself was not disputed as inaccurate or fabricated
- 📌 Dr. Roman's critique is scientifically legitimate: the undetectable PSA in Case 2 is an expected outcome of apalutamide, and the nivolumab response in Case 3 is well within the known efficacy range of PD-1 inhibitors for BRAF-mutated melanoma
- 📌 The breast cancer case (83-year-old, CA 27.29 from 316 → 36.6 in 8 months, 3-year remission) is arguably the most difficult to dismiss, given the multisystem metastatic disease severity and sustained duration — but fulvestrant and radiation were co-administered
- 📌 MDACC data (2026 ASCO abstract) shows 182 cancer patients self-reporting FBZ use at a single institution from 2020–2024, with 138 different dosing schedules — illustrating the scale of uncontrolled self-medication in the absence of clinical guidance
- 📌 What is needed: pharmacokinetic studies, Phase I dose-escalation trials, biomarker identification for patient selection, and ultimately randomized controlled trials before any efficacy claims can be substantiated
- 📌 Until such trials exist, fenbendazole remains an investigational compound with compelling preclinical data and intriguing case-level signals — but no proven efficacy in cancer by current evidence standards
Disclaimer: This article is for educational and informational purposes only. It does not constitute medical advice, diagnosis, or treatment recommendations. Fenbendazole is not approved by any regulatory agency for use in human cancer treatment. All medications, supplements, and therapies discussed here should only be considered under the supervision of a qualified, licensed healthcare professional who is aware of your complete medical history and current treatment regimen.
Scientific References
- Makis W, Baghli I, Martinez P. (2025). Fenbendazole as an Anticancer Agent? A Case Series of Self-Administration in Three Patients. Case Rep Oncol. 18(1):864–871. [Retracted: Case Rep Oncol. 2026;19(1):169] PMC12215191
- Retraction Statement: Paper by William Makis, Ilyes Baghli, and Pierrick Martinez. (2026). Case Rep Oncol. 19(1):169. doi:10.1159/000549387. PMC12823103
- Abughanimeh O, Evans T, Kallam A. (2020). Fenbendazole as a Treatment for Diffuse Large B-Cell Lymphoma. Austin Hematology. Austin Publishing Group
- Phan MV, Willis J, Hu ZI. (2026). A single institutional experience on patterns of ivermectin and fenbendazole use among patients with gastrointestinal cancers. J Clin Oncol. 44(2_suppl):818. ASCO 2026 Abstract
- Dogra N, Kumar A, Mukhopadhyay T. (2018). Fenbendazole acts as a moderate microtubule destabilizing agent and causes cancer cell death by modulating cell cycle arrest, p53 expression and GLUT transporter downregulation. Scientific Reports (Nature), 8, 11926. PMC6098001
- Marchward. (2026, February 25). Correction: Recent Retraction of a Case Study on Fenbendazole Use in Cancer Patients. marchward.com
- Medindia. (2025, December 24). Fenbendazole and Cancer Remission: What a New Case Series Shows. medindia.net
- Roman D. (2026, March 7). Fenbendazole and Cancer: Evidence, Myths & Risks! [YouTube]. Pacific Northwest Urology Specialists. youtube.com
- Karger Publishers. (2025). Retracted Paper — Fenbendazole as an Anticancer Agent? A Case Series of Self-Administration in Three Patients. Case Rep Oncol. 18(1):856–863. karger.com
Shop Sanare Lab
Below are commonly referenced items. Links are provided for convenience — always review the label and consult a professional before use.
180 capsules — 99% purity, laboratory tested
180 capsules — higher dose option
120 capsules — with Black Pepper for absorption
6 / 12 / 18 mg — 100 tablets
Disclaimer: Links are informational and for convenience. This site does not provide medical advice and does not endorse any specific vendor. Always verify product quality, labeling, and consult a licensed professional for health decisions.