Gastrointestinal cancers — colorectal, pancreatic, and hepatocellular — are among the most-discussed cancer types in online fenbendazole communities. On social media forums dedicated to repurposed drug research, questions about FBZ and colon cancer, pancreatic cancer, and liver cancer appear daily, driven in part by the extreme difficulty of treating these cancers with conventional approaches alone. Colorectal cancer is the third most diagnosed cancer worldwide and the second leading cause of cancer death. Pancreatic cancer carries a five-year survival rate under 12%. These grim statistics have driven patients and researchers alike to explore every available avenue, including benzimidazole antiparasitics.
This article synthesizes the preclinical and early clinical evidence around fenbendazole and its benzimidazole relatives in three GI cancer types: colorectal cancer (CRC), pancreatic cancer (PC), and hepatocellular carcinoma (HCC). We cover cell cycle arrest data from a 2022 AACR abstract, the unique finding that FBZ retains activity in 5-fluorouracil-resistant CRC cells via p53-independent ferroptosis, parbendazole + gemcitabine synergy in pancreatic cancer, and the UCSF liver cancer benzimidazole research. We also examine a 2026 safety case report from a metastatic colon cancer patient that carries important lessons about monitoring and dose escalation.
⚠️ Educational Disclaimer
This article is for research and informational purposes only. It does not constitute medical advice and is not a substitute for professional oncology consultation. Always discuss any investigational protocol with your physician before making any treatment decisions.
🔬 Why GI Cancers Are at the Center of FBZ Research
The gastrointestinal tract is a particularly relevant site for benzimidazole pharmacology. Orally administered benzimidazoles like fenbendazole are absorbed from the GI tract, meaning that intestinal and colonic tissues are exposed to relatively higher local concentrations compared to many other tissues. This has led researchers to examine whether these drugs might have particular relevance in GI-originating tumors.
Beyond pharmacokinetics, GI cancers share several biological features that align with FBZ's known mechanisms of action:
- High proliferative rate: Colorectal tumors are driven by rapid epithelial cell turnover, making cell cycle disruption especially relevant.
- Chemotherapy resistance: Both colorectal (5-FU resistance) and pancreatic (gemcitabine resistance) cancers are notorious for developing resistance to first-line agents.
- Metabolic dependency: GI cancers show strong reliance on glucose metabolism and microtubule-driven cell division — two pathways benzimidazoles target directly.
- p53 mutations: Common in colorectal and pancreatic cancers, yet fenbendazole retains activity through p53-independent pathways, which is a significant mechanistic finding.
The Joe Tippens Protocol, which popularized fenbendazole in cancer communities, involved a patient with small cell lung cancer — but the community's attention quickly expanded to GI cancers given their prevalence and unmet need.
🔬 Colorectal Cancer: AACR 2022 Data on Cell Cycle Arrest
One of the most important pieces of evidence for FBZ in colorectal cancer comes from a 2022 abstract presented at the American Association for Cancer Research (AACR) Annual Meeting — one of the most prestigious oncology conferences in the world. The study, by Kang and Kim, specifically investigated fenbendazole in both colorectal cancer cell lines and patient-derived colon cancer organoids — a more clinically relevant model than standard 2D cell cultures (AACR 2022, Abstract 2313).
The researchers specifically chose 3D tumor organoids because standard cell lines fail to recapitulate the physiology of solid tumors. This methodological decision increases the translational relevance of the findings.
Key findings from the AACR 2022 abstract:
- ✅ Fenbendazole induced apoptosis within 24 hours in colon cancer cells, with the effect extending over a longer term.
- ✅ FBZ markedly suppressed proliferation rate via cell cycle arrest — not just slowing growth, but disrupting the cell division machinery.
- ✅ Molecular screening of cell cycle regulators showed drastic downregulation of CDK1 phosphorylated at Tyr15 and cyclin B1, both critical regulators of M phase (mitotic) transition.
- ✅ In an in vivo AOM/DSS colorectal tumor-bearing mouse model, oral FBZ administration reduced not only tumor cell numbers but also lowered tumor grades.
The downregulation of CDK1/cyclin B1 is mechanistically significant. Cyclin B1 is the essential mitotic cyclin — without it, cells cannot progress through mitosis. This explains why FBZ can halt the rapid proliferation that characterizes colorectal cancer tumors.
| Model | Effect | Timeframe | Key Molecular Target |
|---|---|---|---|
| Colon cancer cells (2D) | Apoptosis induction | Within 24 hours | Caspase-3-PARP pathway |
| Patient-derived 3D organoids | Proliferation suppression | Extended time course | CDK1 (Tyr15) + Cyclin B1 |
| AOM/DSS mouse model (oral FBZ) | Reduced tumor number + lower grade | In vivo treatment period | Cell cycle arrest (M phase) |
💊 FBZ and 5-FU-Resistant Colorectal Cancer: The Ferroptosis Angle
Drug resistance is the central challenge in colorectal cancer treatment. 5-fluorouracil (5-FU) remains the mainstay of standard chemotherapy for CRC, but resistance mechanisms — including p53 mutations and altered MAPK signaling — frequently develop, leaving patients with few options.
A 2022 study published in the Korean Journal of Physiology & Pharmacology directly investigated FBZ in 5-FU-resistant SNU-C5 colorectal cancer cells — one of the most clinically relevant resistance models available (PMC9437363). The findings were striking in several respects:
- Fenbendazole outperformed albendazole in 5-FU-resistant cells, making it the preferred benzimidazole in this resistance context.
- FBZ induced both apoptosis and G2/M cell cycle arrest in both sensitive and resistant CRC cells — meaning resistance to 5-FU did not confer cross-resistance to FBZ.
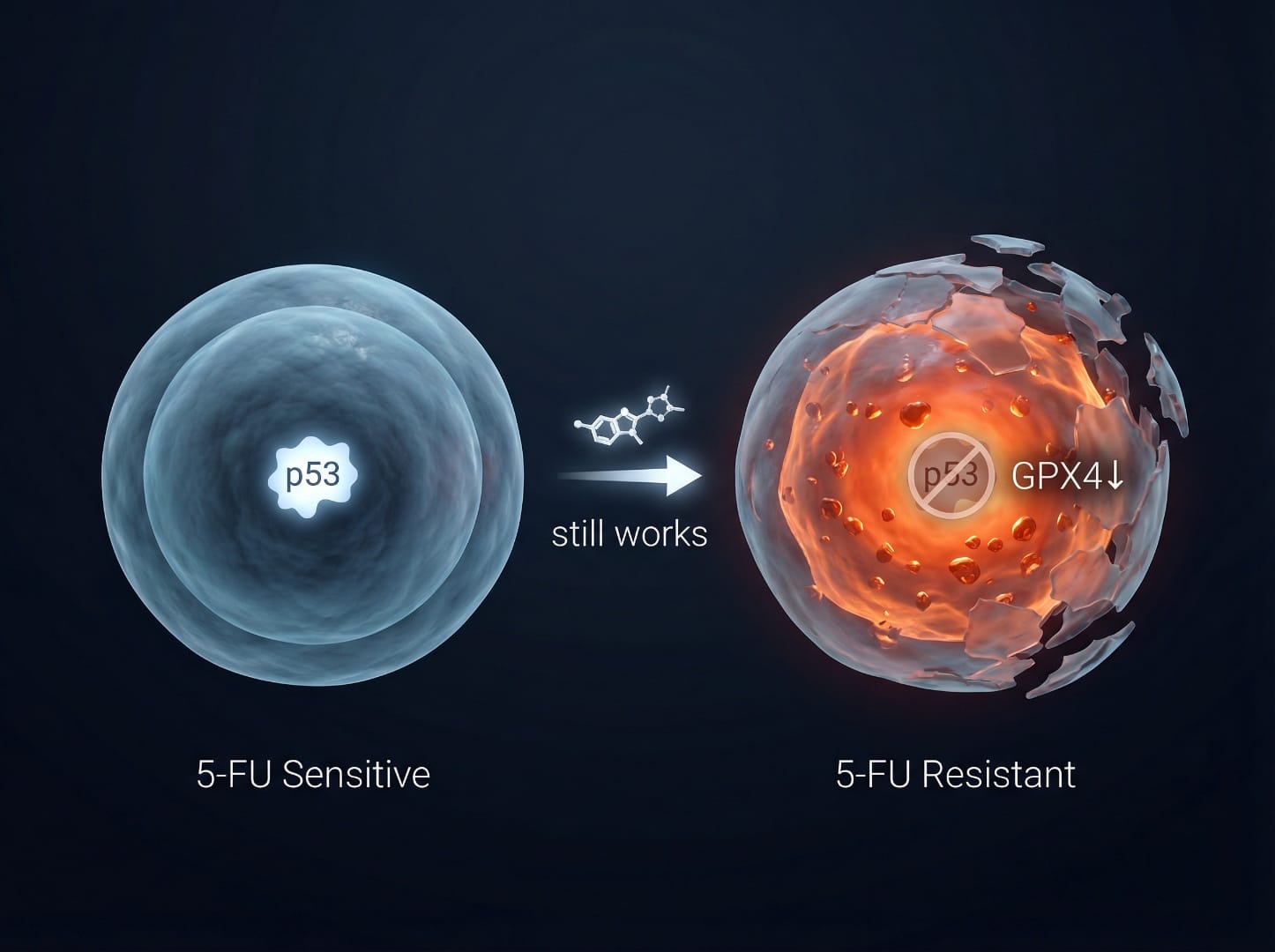
- Critically, in the resistant SNU-C5/5-FUR cells, FBZ triggered apoptosis without activating p53. This is mechanistically important because p53 mutations are a major driver of 5-FU resistance, yet FBZ operates around this barrier.
- The resistant cells showed enhanced ferroptosis — an iron-dependent, non-apoptotic form of cell death — via suppression of GPX4 and SLC7A11. This ferroptosis-augmented apoptosis was actually greater in resistant cells than in 5-FU-sensitive ones.
The Frontiers 2025 pyroptosis paper's discussion section also cites this finding, noting that "in 5-FU-resistant CRC, FBZ induced apoptosis without affecting p53 expression and could enhance p53-independent iron death to promote apoptosis" (Frontiers Pharmacology 2025).
📌 Why this matters: A drug that retains cytotoxic activity in chemotherapy-resistant cells via a completely different mechanism (ferroptosis rather than p53-dependent apoptosis) has genuine therapeutic rationale as an add-on strategy in refractory CRC — though this remains preclinical evidence only.
| Cell Type | FBZ Mechanism | p53 Involvement | Ferroptosis | Notes |
|---|---|---|---|---|
| SNU-C5 (5-FU sensitive) | p53-mediated apoptosis, G2/M arrest | Activated | Partial augmentation | Standard response pathway |
| SNU-C5/5-FUR (resistant) | p53-independent apoptosis + ferroptosis | Not activated | Enhanced (GPX4↓, SLC7A11↓) | Greater ferroptosis than sensitive cells |
🦀 Pancreatic Cancer: Parbendazole + Gemcitabine Synergy
Pancreatic cancer is arguably the most challenging GI malignancy to treat. With a five-year survival rate below 12% and most patients presenting with advanced disease, there is an urgent need for novel treatment approaches. Gemcitabine remains the first-line chemotherapy standard, yet resistance is nearly universal over time.
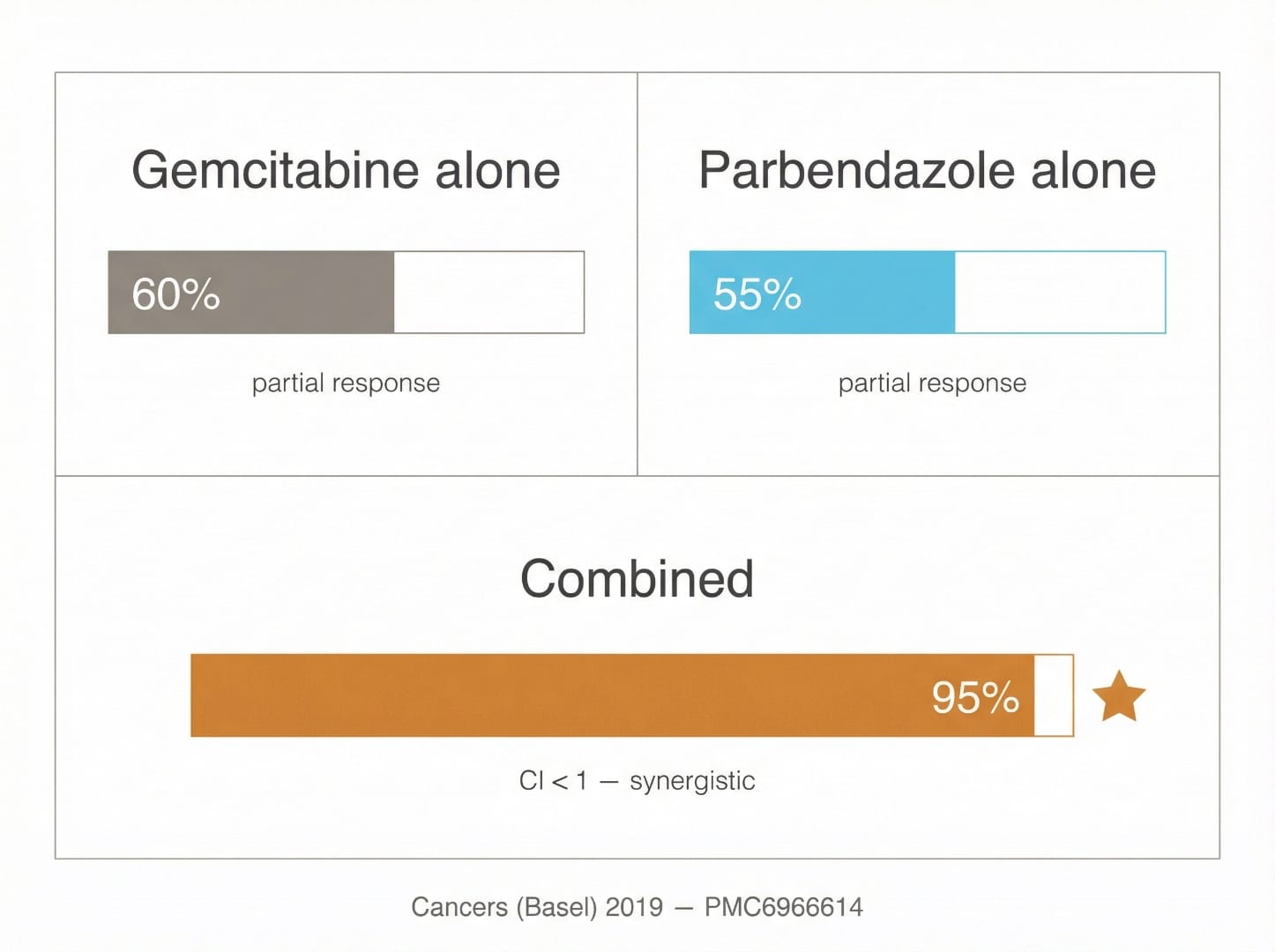
A pivotal 2019 study published in Cancers (Basel) tested all four FDA-approved benzimidazoles — including fenbendazole — against two pancreatic cancer cell lines with distinct genetic backgrounds (AsPC-1 and Capan-2) (PMC6966614). The results established important evidence for the benzimidazole class in pancreatic cancer:
- All four benzimidazoles affected PC cell viability in a dose-dependent fashion, with IC₅₀ values in the nanomolar-to-low-micromolar range — concentrations potentially achievable with standard oral dosing.
- Parbendazole was the most potent of the four, with IC₅₀ values in the nanomolar range — lower than other benzimidazoles including fenbendazole.
- Parbendazole completely abolished clonogenic activity (the ability of cancer cells to self-renew) at even the lowest tested concentrations.
- The drug caused irregular mitotic spindle formation, G2/M cell cycle arrest, mitotic catastrophe, and ultimately apoptosis — the same core microtubule disruption mechanism shared by fenbendazole.
- Most significantly: combinations of parbendazole + gemcitabine showed synergistic (not merely additive) effects on PC cell viability, confirmed by CompuSyn analysis with combination index (CI) < 1.
That fenbendazole is a structural cousin to parbendazole — both are benzimidazole carbamate antiparasitics — means these findings are highly relevant to understanding FBZ's potential class effects. As the Healthline review of FBZ in pancreatic cancer notes, early preclinical evidence for the benzimidazole class shows promise through microtubule disruption and metabolic interference, though human clinical evidence remains absent.
Mechanisms in Pancreatic Cancer Cells
The benzimidazole class targets pancreatic cancer through several complementary pathways:
- Microtubule disruption: Binds β-tubulin, preventing proper mitotic spindle formation. Pancreatic cancer cells require intact microtubules for rapid division and metastatic migration.
- G2/M cell cycle arrest: Trapped in mitosis, cells cannot complete division and undergo mitotic catastrophe.
- DNA damage induction: Parbendazole (and likely related compounds) activates DNA damage response pathways, adding a second route to apoptosis.
- Cell migration impairment: Microtubule disruption also impairs cancer cell movement, potentially reducing metastatic spread.
- WNT/β-catenin signaling inhibition: Benzimidazoles may modulate the WNT pathway, which plays a key role in PC's invasive and immunotolerant behavior.
- Gemcitabine synergy: When combined with gemcitabine, the effect exceeds what either drug achieves alone — a finding with direct clinical relevance.
| Benzimidazole | IC₅₀ in AsPC-1 | IC₅₀ in Capan-2 | Gemcitabine Synergy | Notes |
|---|---|---|---|---|
| Parbendazole | Lowest (nanomolar) | Lowest (nanomolar) | Yes (CI < 1) | Most potent of 4 tested |
| Fenbendazole | Low micromolar | Low micromolar | Not directly tested | Same mechanistic class |
| Mebendazole | Low micromolar | Low micromolar | Not directly tested | Human-formulated alternative |
| Oxibendazole | Low micromolar | Low micromolar | Not directly tested | Less studied in cancer |
For patients exploring pancreatic cancer protocols, see our Fenbendazole Dosage Guide and the ISOM Protocol overview for context on how benzimidazoles are used within structured integrative frameworks.
Products Referenced in This Research
Pharmaceutical-grade fenbendazole referenced in the studies above — 99% purity, lab-tested with full certificates of analysis.
180 capsules — 99% purity, laboratory tested
180 capsules — higher dose option
120 capsules — with Black Pepper for absorption
Disclaimer: For convenience only. Consult a licensed professional before use.
⚠️ Current Limitations and What the Research Cannot Tell Us
As compelling as the preclinical findings are, it is essential to contextualize them honestly. The current body of evidence for FBZ in GI cancers is almost entirely preclinical — conducted in cell cultures and mouse models. Several critical limitations apply:
- No randomized controlled trials: There are currently no published Phase II or Phase III trials evaluating fenbendazole specifically in colorectal, pancreatic, or liver cancer patients.
- Bioavailability challenges: Fenbendazole has poor and highly variable oral bioavailability in humans (approximately 20-40% depending on formulation and food content). Plasma concentrations achievable with standard dosing may differ substantially from the concentrations used in cell culture experiments.
- Organoid models vs. real tumors: Even the 3D organoid models used in the AACR study — while superior to standard cell lines — cannot fully replicate the immune microenvironment, stromal tissue, and vascular supply of human tumors.
- GI cancer heterogeneity: Colorectal cancer alone encompasses dozens of molecular subtypes (MSI-H, MSS, KRAS-mutant, BRAF-mutant, etc.) with different biology and treatment responses. Whether FBZ's efficacy varies across these subtypes is unknown.
- Drug interactions: As the PMC12836008 case illustrates, FBZ can cause hepatotoxicity, which is particularly concerning for GI cancer patients already under metabolic stress — and for those on hepatically metabolized chemotherapy agents.
🩺 Liver and HCC: A Benzimidazole Cousin Shows Promise
Hepatocellular carcinoma (HCC) is the most common primary liver cancer and a frequent complication of cirrhosis. Standard treatment with sorafenib, a tyrosine kinase inhibitor, extends survival but rarely achieves durable remission. Researchers at UCSF used computational data-mining tools to identify a promising combination approach (UCSF 2017).
The UCSF team identified niclosamide ethanolamine (NEN) — a soluble salt of niclosamide, which like fenbendazole is in the broader antiparasitic family used in veterinary medicine — as a candidate for HCC. When combined with sorafenib in a patient-derived HCC tissue engrafted into mouse models, the combination:
- Stopped tumor growth in the experimental model
- Reversed a 274-gene molecular disease signature (comparing cancerous vs. normal liver tissue) after six weeks of treatment
- Targeted multiple genes simultaneously, suggesting it could be effective despite the genetic heterogeneity typical of HCC
Separate from the UCSF work, a 2022 study published in PubMed (PMID 35110505) directly investigated fenbendazole in H4IIE rat hepatocellular carcinoma cells (PubMed 35110505). The findings revealed a critical and nuanced pattern:
- FBZ dose-dependently suppressed growth and induced apoptosis only in actively growing (proliferating) cells
- In fully confluent, quiescent (non-dividing) cells, FBZ showed no significant activity — a finding with important implications
- The mechanism involved p21-mediated cell cycle arrest at both G1/S and G2/M phases, with upregulation of p21 and suppression of cyclin D1 and cyclin B1
- No significant changes were observed in autophagy, cell migration, glycolytic markers, or ROS generation
📌 The quiescent cell finding is clinically significant: It suggests FBZ may have a selective action against actively dividing cancer cells while sparing quiescent normal liver cells — a desirable therapeutic profile. However, it also implies that slowly growing or dormant tumors may be less responsive.
GI Cancer Research Summary Table
| Cancer Type | Key Study | Drug | Model | Key Finding | Evidence Level |
|---|---|---|---|---|---|
| Colorectal (CRC) | AACR 2022 (Kang & Kim) | Fenbendazole | Cell line + 3D organoids + mouse | Apoptosis in 24h; CDK1/cyclin B1 ↓; tumor grade ↓ | Preclinical |
| Colorectal (5-FU resistant) | Park et al. 2022 (PMC9437363) | Fenbendazole | SNU-C5/5-FUR cell line | p53-independent apoptosis + ferroptosis augmentation | Preclinical |
| Pancreatic | Florio et al. 2019 (PMC6966614) | Parbendazole (benzimidazole class) | AsPC-1 + Capan-2 cell lines | Nanomolar activity; synergy with gemcitabine (CI < 1) | Preclinical |
| Hepatocellular (HCC) | Park 2022 (PMID 35110505) | Fenbendazole | H4IIE rat HCC cells | p21-mediated G1/S + G2/M arrest; active cells only | Preclinical |
| Hepatocellular (HCC) | UCSF 2017 | Niclosamide ethanolamine (benzimidazole cousin) | Patient-derived xenograft mouse | Tumor growth arrest; reversed 274-gene signature with sorafenib | Preclinical |
🩺 The 2026 Case Report: A Safety Lesson for Colon Cancer Patients
In January 2026, a case report published in the World Journal of Clinical Cases described a 47-year-old woman with metastatic colon cancer (Lynch syndrome variant) who had been self-administering fenbendazole alongside her oncology treatment (PMC12836008). This case is worth examining carefully because it illustrates several safety principles directly relevant to GI cancer patients.
Case Details
- The patient was on nivolumab/relatlimab (Opdualag) for 9 months — a dual immune checkpoint inhibitor regimen — with her last infusion 4 weeks before admission.
- She began self-administering fenbendazole (Panacur C) after consulting a "holistic healer" and being influenced by social media "fenben protocols."
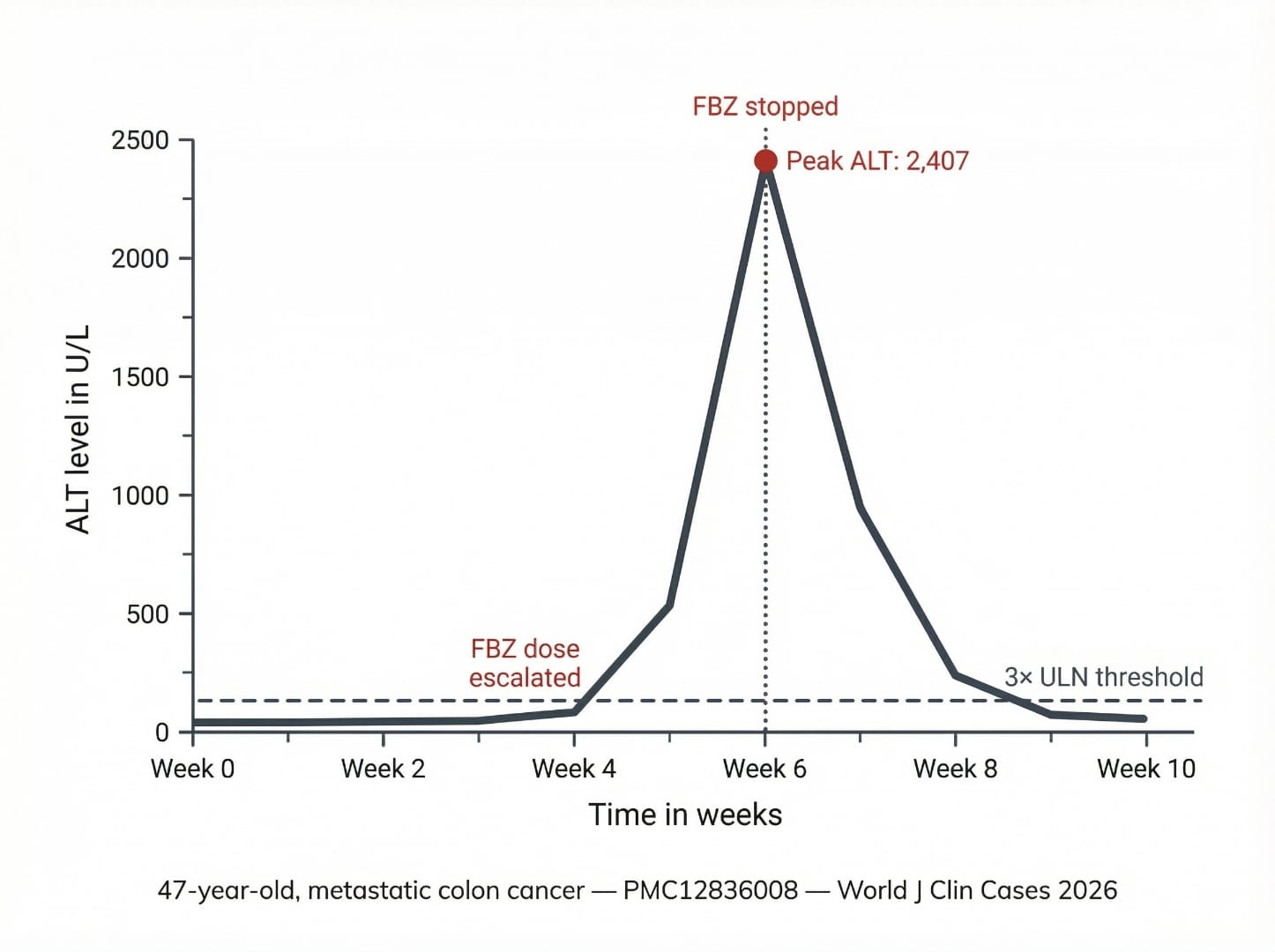
- She started at 222 mg three times weekly for 6 weeks, then escalated to 222 mg daily (approximately 1,554 mg weekly total) in the two weeks before presentation.
- She presented with 1 week of nausea, fatigue, dark urine, and jaundice.
- Labs showed severe hepatocellular injury: AST 2,435 U/L, ALT 2,407 U/L, elevated bilirubin, INR 1.52 — a hepatocellular pattern.
- RUCAM causality score of 8 = probable FBZ-induced liver injury.
- All other causes (viral hepatitis, autoimmune, acetaminophen, infection) were ruled out.
- After stopping FBZ, ALT dropped 78% within 10 days and normalized fully — confirming FBZ as the causative agent.
- Importantly, the patient successfully resumed immunotherapy (nivolumab/relatlimab) after FBZ discontinuation with no recurrence of liver injury.
Key Safety Lessons from This Case
- Dose escalation is high-risk: The liver injury onset correlated with dose increase to daily 222 mg — a pattern seen in other FBZ DILI cases. Gradual escalation without monitoring is particularly dangerous.
- Concurrent immunotherapy compounds the risk: Immune checkpoint inhibitors (ICIs) can cause immune-mediated hepatitis that looks identical to drug-induced liver injury. In this patient, the diagnostic challenge was distinguishing FBZ-DILI from ICI hepatitis — an important distinction because the treatments differ significantly.
- GI cancer patients have compromised liver reserves: Metastatic colon cancer frequently involves liver metastases. Any patient with known or suspected hepatic involvement has reduced capacity to tolerate hepatotoxic insults.
- Disclose to your oncologist: The diagnostic complexity in this case was substantially increased because the FBZ use was undisclosed. Early disclosure enables proper monitoring and safer decision-making.
⚠️ This case is discussed in much greater depth in our dedicated post on Fenbendazole Liver Safety and Side Effects.
💊 Dosing Considerations for GI Cancer Patients
Patients with GI cancers face unique pharmacological considerations when exploring FBZ. The following reflects published protocols and known pharmacology — not medical advice.
Absorption and GI Context
Fenbendazole is a highly lipophilic compound with poor aqueous solubility. Its oral bioavailability is significantly enhanced when taken with a fatty meal. For GI cancer patients:
- Always take with food containing fat (e.g., olive oil, avocado, nuts, fatty fish). Studies show absorption can increase 3-5x with food vs. fasting.
- GI cancer patients who have undergone bowel resection, colostomy, or have significant mucosal damage may have altered drug absorption — making plasma level monitoring especially relevant.
- Pancreatic cancer patients with exocrine insufficiency may have impaired fat digestion, potentially reducing FBZ absorption further. Pancreatic enzyme supplementation may affect this.
- Patients with liver metastases or compromised hepatic function have reduced capacity to metabolize FBZ and may be at higher risk of drug accumulation and hepatotoxicity.
Standard Reference Dosing (Educational Only)
| Protocol Reference | Dose | Schedule | Notes for GI Cancer Patients |
|---|---|---|---|
| Joe Tippens / common community protocols | 222 mg/day | 7 days on / off, or continuous | Start with 3x/week before daily dosing; monitor LFTs |
| Research lab doses (preclinical) | ~1 mg/kg (mouse, oral) | Daily | Human equivalent dosing not established |
| Higher dose protocols | 444 mg/day | 5 days on / 2 days off | Higher liver monitoring frequency required |
| Post-liver injury restart (if any) | Lower dose, slower titration | Medical supervision only | Only under oncologist guidance; check LFTs at 2–4 weeks |
See the complete Fenbendazole Dosage Guide for detailed protocol schedules, and our comparison of Fenbendazole vs. Ivermectin for those exploring combinations.
Liver Monitoring Protocol for GI Cancer Patients
Because GI cancers can directly involve or metastasize to the liver, any FBZ user with GI cancer should apply a more rigorous monitoring schedule than the general population:
- 📌 Baseline LFTs before starting (AST, ALT, ALP, bilirubin, GGT, albumin)
- 📌 Repeat at 2 weeks after initiation or dose change
- 📌 Monthly monitoring during stable dosing
- 📌 Immediate testing if any symptoms appear: fatigue, nausea, dark urine, jaundice, right upper quadrant pain
- 📌 Stop FBZ immediately if ALT/AST exceeds 3× upper limit of normal (ULN) and consult physician
- 📌 If on immune checkpoint inhibitors (pembrolizumab, nivolumab, relatlimab, etc.), always disclose FBZ use — distinguishing FBZ-DILI from ICI hepatitis requires knowing the full medication history
🔗 How FBZ Fits into GI Cancer Protocols
Within structured integrative oncology frameworks, fenbendazole is not typically used as a standalone therapy but as part of a multi-agent metabolic approach. The ISOM Protocol, developed by researchers affiliated with the International Society for Orthomolecular Medicine, positions benzimidazoles alongside metabolic agents (metformin, repurposed drugs), orthomolecular support (vitamin D3, vitamin C, zinc, curcumin), and dietary interventions (ketogenic diet, intermittent fasting).
For GI cancer patients specifically, several combination considerations arise:
- With standard chemotherapy: The parbendazole + gemcitabine synergy data (PMC6966614) provides preclinical rationale for benzimidazole + gemcitabine combination in pancreatic cancer, though no human trial has yet validated this. See our full post on Fenbendazole and Chemotherapy.
- With curcumin: Curcumin has well-established anti-inflammatory and NF-κB inhibitory effects in CRC, and its combination with FBZ is part of several community protocols. Curcumin may also provide some hepatoprotective support. Absorption with black pepper (piperine) is essential.
- With vitamin D3: Vitamin D deficiency is associated with increased colorectal cancer risk and worse outcomes; optimal D3 levels (40–80 ng/mL) support immune function alongside any investigational protocol.
📊 Comparing Available Evidence Across GI Cancers
Not all GI cancers have equal research depth when it comes to benzimidazole data. Here is a transparent comparison of the current evidence landscape:
| Cancer Type | Number of Preclinical Studies | Human Case Reports | Clinical Trials | Strongest Finding | Research Maturity |
|---|---|---|---|---|---|
| Colorectal Cancer | Multiple (cell lines, organoids, mouse models) | Limited (indirect via case series) | None specific to FBZ | 24h apoptosis; overcomes 5-FU resistance via ferroptosis | ⭐⭐⭐ Moderate |
| Pancreatic Cancer | Limited (parbendazole class data) | None published | None | Nanomolar activity; gemcitabine synergy (CI < 1) | ⭐⭐ Early |
| Hepatocellular Carcinoma | Limited (FBZ in H4IIE cells; NEN in PDX mouse) | None published | None | p21-mediated arrest; active cells selectively targeted | ⭐⭐ Early |
| Gastric Cancer | Very limited (GSDME pathway data) | None | None | GSDME-mediated pyroptosis theoretical basis | ⭐ Very early |
📌 Key Takeaways
- ✅ The AACR 2022 abstract demonstrated fenbendazole induces apoptosis in colorectal cancer cells and patient-derived organoids within 24 hours, via CDK1/cyclin B1 downregulation.
- ✅ In 5-FU-resistant colorectal cancer, FBZ retains cytotoxic activity through a p53-independent mechanism involving enhanced ferroptosis — directly relevant to refractory CRC.
- ✅ Parbendazole (benzimidazole cousin) showed nanomolar IC₅₀ values in pancreatic cancer cells and synergized with gemcitabine in preclinical models — the best benzimidazole class evidence for pancreatic cancer.
- ✅ In hepatocellular carcinoma, FBZ caused p21-mediated cell cycle arrest selectively in proliferating cells, with the UCSF research highlighting benzimidazole class + sorafenib synergy in HCC mouse models.
- ⚠️ A 2026 case report (PMC12836008) of a 47-year-old colon cancer patient on immunotherapy who developed severe FBZ-induced liver injury highlights the critical importance of liver monitoring, disclosure to oncologists, and avoiding unsupervised dose escalation.
- 📌 GI cancer patients have uniquely elevated liver risk from FBZ due to potential liver metastases, concurrent hepatotoxic therapies, and altered hepatic metabolism — requiring more rigorous LFT monitoring than other cancer types.
- 📌 Absorption optimization (always with fatty food) is especially relevant for GI cancer patients, whose surgeries and disease processes may alter drug absorption in ways not applicable to the general population.
- 📌 All current evidence is preclinical. Fenbendazole is not approved or validated for any human cancer treatment. Human trials are urgently needed.
Disclaimer: This article is for educational and research purposes only. It does not constitute medical advice. Fenbendazole is not approved for cancer treatment in humans. All treatment decisions — including the use of any investigational or repurposed compounds — must be made under the supervision of a qualified oncology professional.
Scientific References
- Kang K-S, Kim D-H. (2022). Fenbendazole induces cell cycle arrest in colorectal cancer cells and patient-derived colon cancer organoids. AACR Annual Meeting 2022. Cancer Res 82(12_Suppl):Abstract nr 2313. AACR 2022 Abstract 2313
- Park D-H et al. (2022). Anti-cancer effects of fenbendazole on 5-fluorouracil-resistant colorectal cancer cells. Korean Journal of Physiology & Pharmacology, 26(5), 377–388. PMC9437363
- Florio R et al. (2019). The benzimidazole-based anthelmintic parbendazole: A repurposed drug candidate that synergizes with gemcitabine in pancreatic cancer. Cancers (Basel), 11(12), 2042. PMC6966614
- Park D. (2022). Fenbendazole suppresses growth and induces apoptosis of H4IIE hepatocellular carcinoma cells via p21-mediated cell cycle arrest. PubMed PMID 35110505. PubMed 35110505
- Krishnan A, Lucas K, Maas L, Woreta TA. (2026). Differentiating fenbendazole-induced liver injury from immune checkpoint inhibitor hepatitis in a patient with metastatic colon cancer. World Journal of Clinical Cases, January 2026. PMC12836008
- UCSF News. (2017). Deworming pill may be effective in treating liver cancer. University of California, San Francisco. UCSF 2017
- Pan T et al. (2025). Fenbendazole induces pyroptosis in breast cancer cells via the caspase-3/GSDME pathway. Frontiers in Pharmacology. Frontiers Pharmacology 2025
- Ruwa R. (2023). Fenbendazole for Pancreatic Cancer: What Research Shows. Healthline. Healthline — Fenbendazole Pancreatic Cancer
Shop Sanare Lab
Below are commonly referenced items. Links are provided for convenience — always review the label and consult a professional before use.
180 capsules — 99% purity, laboratory tested
180 capsules — higher dose option
120 capsules — with Black Pepper for absorption
6 / 12 / 18 mg — 100 tablets
Disclaimer: Links are informational and for convenience. This site does not provide medical advice and does not endorse any specific vendor. Always verify product quality, labeling, and consult a licensed professional for health decisions.