Curcumin, a naturally occurring polyphenol found in turmeric (Curcuma longa), has emerged as one of the most researched plant-derived compounds in modern biomedical science. With over 14,000 published studies indexed on PubMed and dozens of active clinical trials, it is the most intensively studied dietary polyphenol in the context of cancer. A major 2024 review published in Frontiers in Oncology examined a broad body of laboratory, animal, and early-stage human studies to assess curcumin's potential relevance in cancer research, highlighting its wide-ranging biological effects and therapeutic promise.
⚠️ Educational Disclaimer
This article is for research and informational purposes only. It does not constitute medical advice. Do not self-prescribe. Always consult a qualified healthcare provider before using any supplement, especially alongside cancer treatment.
Cancer burden and the search for better-tolerated strategies
Cancer remains the second leading cause of death globally. In 2020 alone, approximately 18.1 million new cancer cases and nearly 10 million cancer-related deaths were reported worldwide. Projections suggest global cancer incidence could increase by 31–47% by 2030–2040, depending on region and cancer type.
While conventional treatments such as chemotherapy, radiation, and targeted therapies have improved survival in many cancers, they are often limited by toxicity, resistance, and long-term side effects. This has driven growing scientific interest in adjunct compounds that may modulate cancer-related pathways with lower toxicity profiles — including curcumin.
🔬 How curcumin affects cancer cells in laboratory models
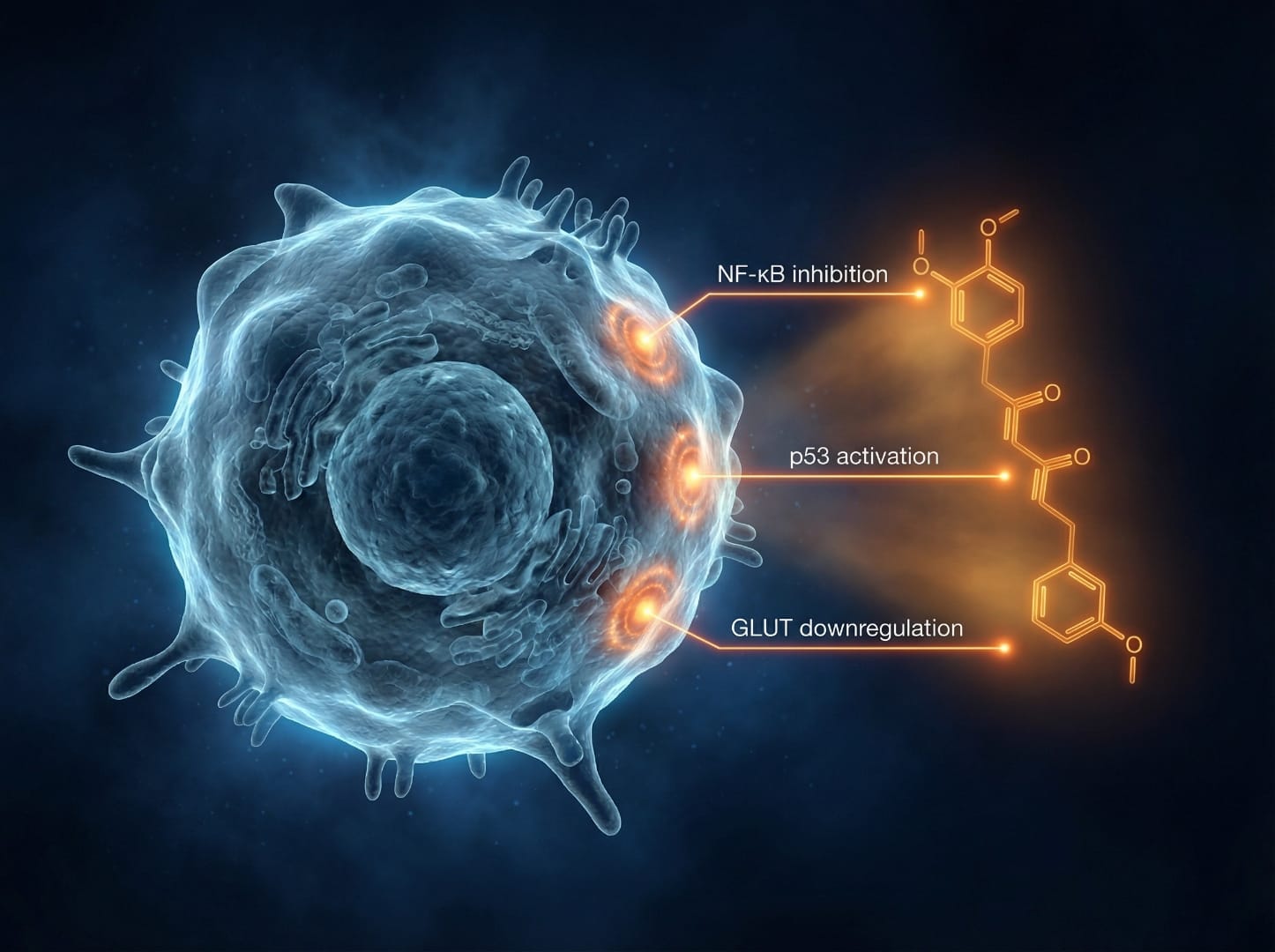
According to the 2024 review, curcumin demonstrates anti-proliferative and pro-apoptotic effects across a wide range of cancer cell lines in laboratory and animal models. These effects are not limited to a single mechanism — curcumin simultaneously targets multiple pathways that are commonly dysregulated in cancer:
| Signaling Pathway | Role in Cancer | Effect of Curcumin |
|---|---|---|
| PI3K / Akt / mTOR | Cell survival and growth | Inhibits Akt phosphorylation, reduces mTOR signaling → G1 arrest |
| NF-κB | Inflammation and cancer cell survival | Directly suppresses NF-κB DNA binding activity |
| JAK / STAT3 | Tumor progression and immune evasion | Blocks STAT3 phosphorylation and nuclear translocation |
| Wnt / β-catenin | Tumor growth and metastasis | Downregulates β-catenin, cyclin D1 and c-Myc |
| MAPK (ERK, JNK, p38) | Proliferation and stress responses | Modulates ERK1/2, activates JNK-mediated apoptosis |
| Hedgehog / Notch | Cancer stem cell maintenance | Reduces Gli activity and Notch1 expression |
By modulating all these pathways simultaneously, curcumin has been shown in experimental models to reduce cancer cell proliferation, induce programmed cell death (apoptosis), inhibit angiogenesis, and suppress invasion and metastasis-related signaling.
🧬 Specific cancer types: what does the preclinical data say?
Curcumin has been studied across a remarkably wide range of cancer types. Here is a summary of key findings from recent reviews:
| Cancer Type | Dosage (in vitro) | Key Mechanism |
|---|---|---|
| 🩺 Prostate cancer | 10–25 μM | Induces apoptosis, inhibits androgen receptor signaling, suppresses PI3K/Akt/mTOR |
| 🩺 Breast cancer | 10–50 μM | Blocks MAPK and NF-κB, restricts proliferation, suppresses Hedgehog/Gli |
| 🩺 Colorectal cancer | 65–125 μM | Targets CDK2–c-MYC–PTBP1 axis, induces G1 arrest, promotes autophagy |
| 🩺 Pancreatic cancer | 20–50 μM | Inhibits NF-κB and STAT3, blocks Notch1 and TGF-β signaling |
| 🩺 Brain cancer (glioma) | 10–50 μM | Induces apoptosis, suppresses PI3K/Akt, crosses BBB in liposomal form |
| 🩺 Liver cancer | 10–30 μM | Inhibits JAK/STAT pathways, induces dose-dependent apoptosis |
| 🩺 Lung cancer (NSCLC) | Varies | Inhibits COX-2 and NF-κB, 36% reduction in intralung tumor weight (in vivo) |
A 2025 study from Frontiers in Oncology identified a new mechanism in colorectal cancer: curcumin directly binds to and inhibits CDK2, disrupting the CDK2–c-MYC–PTBP1 regulatory axis. In xenograft mouse models, curcumin at 100 mg/kg daily significantly reduced tumor volumes and decreased tumor cell density with no visible signs of systemic toxicity.
💊 Bioavailability: the critical challenge
Despite strong laboratory data, curcumin faces a critical limitation: extremely low oral bioavailability.
The numbers tell the story clearly:
- Even doses of 10–12 grams per day produce blood concentrations below 50 nanomolar
- A 2 g oral dose results in serum levels that are often undetectable by standard HPLC analysis
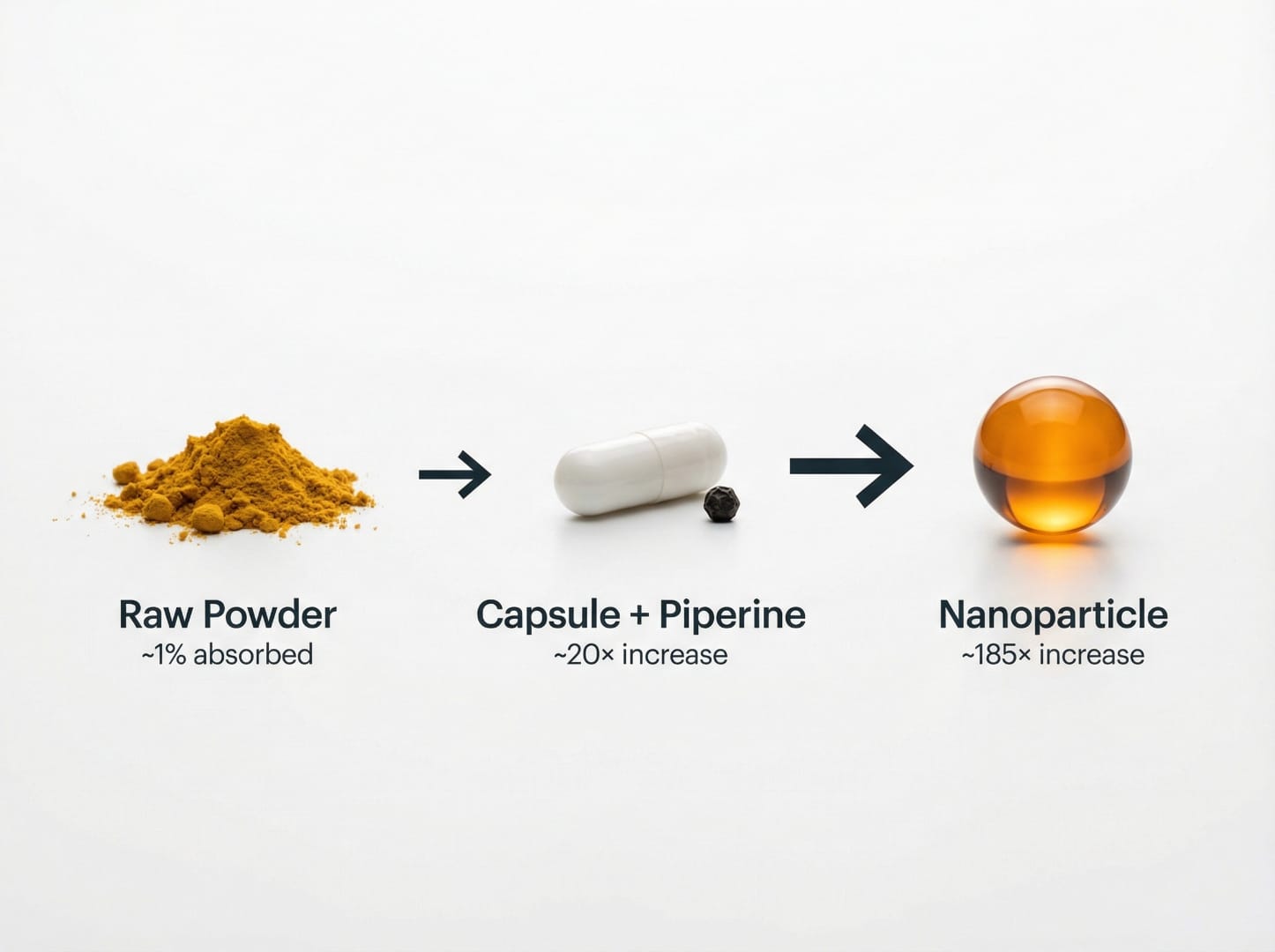
- Systemic bioavailability of raw curcumin is estimated at ~1% or less
- Phase I trials confirmed curcumin is safe even at 12 g/day, but bioavailability remains poor
This explains a well-known paradox: curcumin shows potent effects in cell cultures but far weaker effects in human studies when used in its unformulated (raw powder) form.
🧪 Why formulations matter: phytosomes, piperine, and nanoparticles
To address bioavailability challenges, modern research focuses heavily on curcumin delivery systems. The differences are dramatic:
| Formulation | Bioavailability vs. Raw Curcumin | Notes |
|---|---|---|
| Raw curcumin powder | Baseline (1×) | ~1% systemic absorption, rapidly metabolized |
| Curcumin + Piperine (BioPerine) | ~20× increase | Inhibits liver enzymes slowing metabolism; caution with Rx drugs |
| Phytosome (Meriva) | ~29× increase | Phosphatidylcholine binding, most robust clinical evidence |
| Liposomal curcumin | 5–10× increase | Better tissue delivery (liver, spleen, brain); more expensive |
| Nanoparticles (PLGA) | ~9× higher than curcumin+piperine | Sustained-release profile: 24% at 24h, 43% over 20 days |
| CHC formulation | ~49× increase | Hydrophilic carrier + cellulosic derivative + antioxidants |
| LPS nanocarriers | ~130× increase | Lipopolysaccharide nanocarrier system (preclinical) |
In animal models, nano-formulated curcumin has been shown to reduce tumor size more effectively than free curcumin, sometimes within days rather than weeks, and at much lower doses. According to a 2023 ACS Omega review, the overarching goal of all strategies is to increase curcumin's solubility and bioavailability, since raw curcumin produces undetectable plasma levels even at gram-scale doses.
Curcumin with Enhanced Bioavailability
Below is a curcumin supplement formulated with Black Pepper extract (piperine) to enhance absorption. Link is provided for convenience — always review the label and consult a professional before use.
120 capsules — with Black Pepper & Phytosome Extract for enhanced absorption
🩺 What do human clinical trials show?
While human data remains limited compared to preclinical research, several Phase II randomized controlled trials have now produced concrete results. Here is a summary of the most significant findings:
Breast cancer — curcumin + paclitaxel (150 patients, Armenia)
A Phase II RCT gave 75 patients IV curcumin (CUC-01, 300 mg/week) alongside paclitaxel for 12 weeks. Results:
- Objective response rate at 16 weeks: 50.7% (curcumin group) vs. 33.3% (placebo) — statistically significant (p<0.05)
- At 24 weeks: 29% response in the curcumin group vs. 20% in placebo
- A separate study reported that 300 mg/day curcumin nanoparticles for 6 months produced tumor shrinkage in 60% of participants
Colorectal cancer — curcumin + FOLFOX (27 patients, UK)
A Phase IIa RCT in previously untreated Stage IV colorectal cancer patients added 2 g oral curcumin C3 complex to standard FOLFOX ± bevacizumab:
- 6-month overall survival (per-protocol): 93.3% (curcumin) vs. 55.6% (placebo)
- 6-month progression-free survival: 73.3% vs. 33.3%
- Median overall survival: 596 days (curcumin) vs. 200 days (placebo)
- Objective response at 12 cycles: 53.3% vs. 11.1%
Prostate cancer — curcumin during ADT withdrawal (97 patients, South Korea)
Patients with Stage IV prostate cancer received 1,440 mg/day curcumin for 6 months during androgen deprivation therapy (ADT) withdrawal. The results showed:
- PSA progression at 6 months: 10.3% (curcumin) vs. 30.2% (placebo)
- Adverse events: 15.6% (curcumin) vs. 34.8% (placebo) — curcumin was better tolerated
A separate radiation therapy study in 180 prostate cancer patients found that 3 g/day curcumin markedly reduced PSA levels and decreased radiation-related side effects (inflammation, urological complications) over 3 months.
Multiple myeloma — curcumin + MP regimen (33 patients, Indonesia)
A pilot RCT combined BCM-95 CG curcumin (8 g/day) with melphalan and prednisone. Among patients who completed the 4-month follow-up:
- Remission rate: 75% (curcumin group) vs. 33.3% (placebo)
Oral leukoplakia — curcumin alone (223 patients, India)
A Phase IIb RCT tested 3.6 g/day BCM-95 CG curcumin for 6 months in patients with confirmed oral leukoplakia (a pre-cancerous condition):
- Clinical response: 67.5% (curcumin) vs. 55.3% (placebo)
- Combined clinical + histologic response: 58% vs. 44.6%
Summary of clinical trial data
| Cancer Type | Trial Size | Curcumin Dose | Key Finding |
|---|---|---|---|
| Breast (advanced) | 150 pts | 300 mg IV/week | ORR: 50.7% vs 33.3% with paclitaxel |
| Colorectal (Stage IV) | 27 pts | 2 g oral/day | Median OS: 596 vs 200 days with FOLFOX |
| Prostate (Stage IV) | 97 pts | 1.44 g oral/day | PSA progression: 10.3% vs 30.2% |
| Multiple myeloma | 33 pts | 8 g oral/day | Remission: 75% vs 33.3% with MP |
| Oral leukoplakia | 223 pts | 3.6 g oral/day | Clinical response: 67.5% vs 55.3% |

⚠️ Important context: These are early-phase trials with relatively small sample sizes. The evidence is promising but not yet conclusive for any cancer type. Larger Phase III trials are needed to confirm these findings. Different curcumin formulations, dosages, and administration routes make direct comparisons difficult.
🔬 Curcumin + chemotherapy: synergy data
One of the most active areas of curcumin research is its potential to enhance the effectiveness of standard chemotherapy while reducing side effects. A comprehensive review in Molecules documented synergistic effects across multiple drug combinations:
| Chemotherapy Drug | Cancer Type | Synergistic Effect with Curcumin |
|---|---|---|
| 5-Fluorouracil (5-FU) | Colorectal, Gastric | Overcomes 5-FU drug resistance, reverses multidrug resistance in MMR-deficient cells |
| Doxorubicin | Hodgkin lymphoma | 79% reduction in cell proliferation (vs. doxorubicin alone); reduced tumor weight to 56.5% |
| Cisplatin | Bladder, Ovarian | Potent synergistic effect via caspase-3 activation; reduced tumor volume in mouse models |
| Docetaxel | Prostate, Breast | Enhanced efficacy via COX-2, p53, NF-κB modulation; reduced CEA tumor marker |
| Gemcitabine | Pancreatic, Bile duct | Overcomes gemcitabine resistance; lowest tumor growth rate in combination group (265% vs control) |
| Paclitaxel | Breast | ORR increased from 33.3% to 50.7% (Phase II, 150 patients) |
A 2024 study in Nature Scientific Reports showed that curcumin synergistically enhanced gemcitabine efficacy against gemcitabine-resistant bile duct cancer cells by targeting the LAT2/glutamine pathway. In xenograft mouse models, the combination treatment produced the lowest tumor growth rate of all groups tested.
Notably, curcumin also appears to reduce the side effects of chemotherapy. The combination of curcumin and doxorubicin suppressed the reduction of glutathione peroxidase activity and decreased lipid peroxide levels in the heart — suggesting potential cardioprotective effects during chemotherapy.
🛡️ Curcumin and immunotherapy
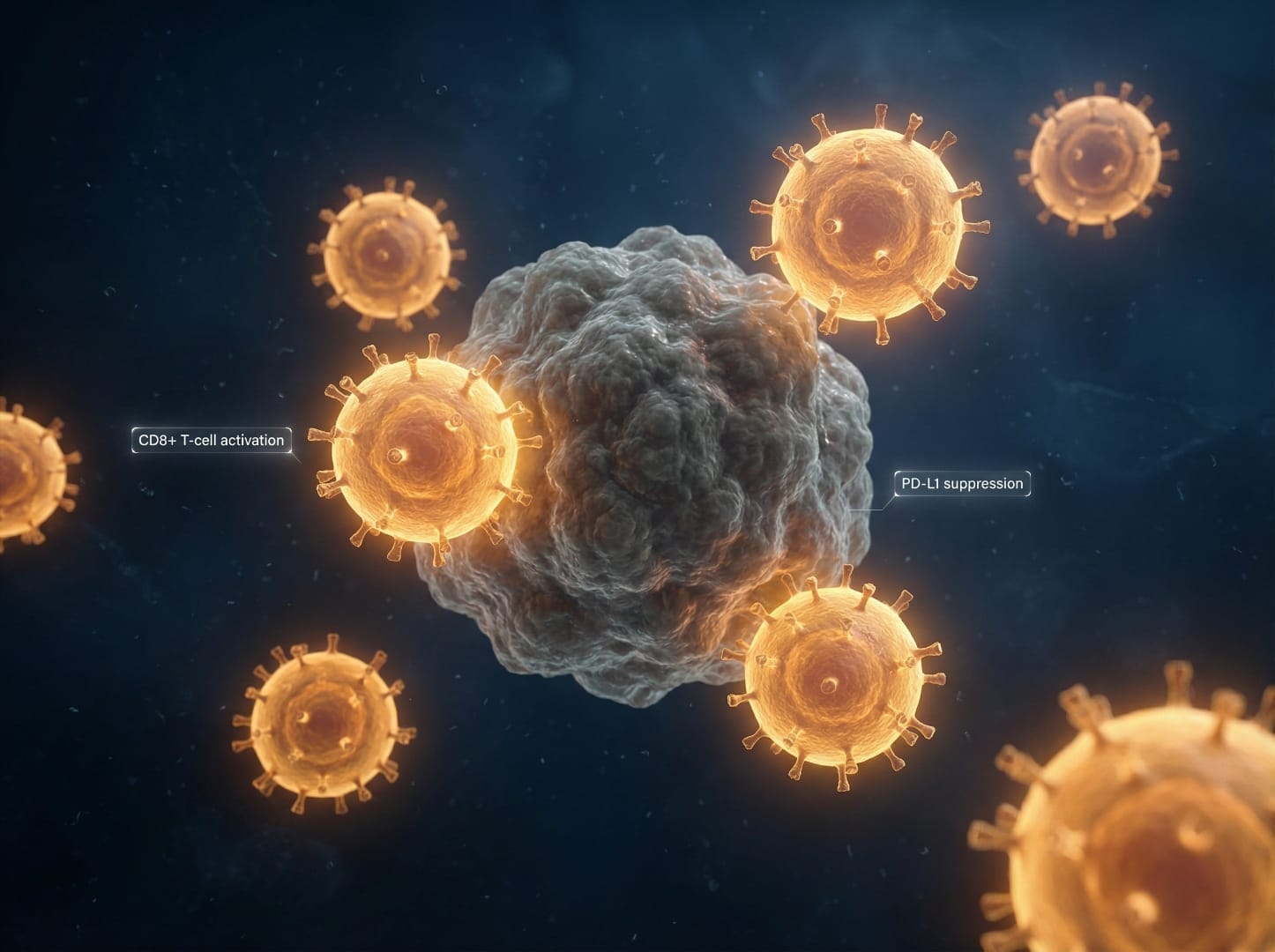
Emerging research has also explored combining curcumin with modern immunotherapy agents. Recent data showed that bisdemethoxycurcumin (BDMC), a curcumin derivative:
- Increased levels of tumor-infiltrating CD8+ T cells
- Enhanced IFN-γ secretion in the blood
- Decreased the number of tumor-infiltrating MDSCs (immune-suppressive cells)
- When combined with anti-PD-L1 antibodies, further enhanced secretion of IFN-γ, granzyme B, and perforin from CD8+ T cells
This is particularly relevant given the growing importance of immune checkpoint inhibitors in cancer treatment. Curcumin's ability to modulate the tumor microenvironment may make it a useful adjunct to PD-1/PD-L1 therapy.
🧬 Oral cancer: a Phase II breakthrough
One of the most promising clinical programs for curcumin is in oral cancer. Led by Dr. Marilene B. Wang at UCLA, a Phase II trial is investigating APG-157, a full-spectrum turmeric pastille, in patients with oral and oropharyngeal cancers.
The preceding Phase I trial confirmed:
- Safety and tolerability of the turmeric pastille
- Significant reduction of inflammatory cytokines in saliva
- Decreased tumor-associated microbes
- Recruitment of T-cells to the tumor microenvironment
APG-157 is expected to induce apoptosis in tumor cells, thereby reducing tumor burden and enhancing outcomes from subsequent local therapy. The Phase II trial is evaluating pathological responses, immune response biomarkers, and changes within tumor tissue.
✅ What this means for informed consumers
From a scientific perspective, the current evidence supports understanding curcumin as:
- A biologically active polyphenol with well-documented molecular effects across at least 7 cancer types
- A compound where formulation quality matters far more than raw dose — phytosome and nanoparticle forms are 9–130× more bioavailable than raw powder
- A promising adjunct to chemotherapy with synergistic effects documented for 5-FU, cisplatin, doxorubicin, docetaxel, gemcitabine, and paclitaxel
- A compound with a favorable safety profile — safe at doses up to 12 g/day in Phase I trials, and associated with fewer adverse events than placebo in at least one large RCT
- An ingredient whose clinical evidence is encouraging but still preliminary — larger Phase III trials are needed
Modern research does not support simplistic claims, but it does justify continued interest in high-quality, bioavailable curcumin formulations as part of evidence-informed wellness strategies.
Bottom line
Curcumin is one of the most extensively studied natural compounds in cancer-related research. While it is not a standalone cancer treatment, the clinical trial data is increasingly compelling: median survival tripled in a colorectal cancer trial, objective response rates nearly doubled in breast cancer when combined with paclitaxel, and PSA progression was reduced threefold in prostate cancer.
The field has moved well beyond "lab-only" findings. With multiple Phase II trials complete and more underway, understanding the difference between raw turmeric, standardized extracts, and advanced delivery systems is more important than ever.
Disclaimer: This content is for educational and research purposes only. It does not constitute medical advice. Always discuss any supplementation with your treating physician before use.
Scientific References
- Ameer SF et al. (2024). Curcumin as a novel therapeutic candidate for cancer: can this natural compound revolutionize cancer treatment? Frontiers in Oncology. DOI: 10.3389/fonc.2024.1438040
- Yuan W et al. (2025). Curcumin inhibits colorectal cancer progression by targeting PTBP1. Frontiers in Oncology. DOI: 10.3389/fonc.2025.1566889
- Howells LM et al. (2019). Phase IIa randomized trial of curcumin with FOLFOX in colorectal cancer. Cancer Prev Res. Review in Pharmaceutics
- Saghatelyan T et al. (2020). Curcumin with paclitaxel in advanced breast cancer: Phase II RCT. Phytomedicine. Review in Pharmaceutics
- Choi YH et al. (2019). Curcumin supplementation during ADT withdrawal in prostate cancer. Urol Oncol. Review in Pharmaceutics
- Santosa D et al. (2020). Curcumin BCM-95 CG with MP regimen in multiple myeloma. Pilot RCT. Review in Pharmaceutics
- Sbrini G et al. (2023). Exploring the contribution of curcumin to cancer therapy: systematic review. Pharmaceutics. PMC10144810
- Khayat MT et al. (2019). Curcumin combination chemotherapy: the implication and efficacy in cancer. Molecules. PMC6680685
- Jantawong C et al. (2024). Curcumin synergistically enhances gemcitabine efficacy in cholangiocarcinoma. Nature Scientific Reports. DOI: 10.1038/s41598-024-66945-7
- Mughal T et al. (2026). Anticancer molecular mechanisms of curcuminoids. Food Sci Nutr. PMC12865664
- Ahmed S et al. (2023). Curcumin formulations for better bioavailability. ACS Omega. PMC10061533
Protocol Stack (Quick Links)
Below are commonly referenced items mentioned in this article. Links are provided for convenience — always review the label and consult a professional before use.
360 mg per capsule — 120 capsules with Black Pepper & Phytosome Extract
180 capsules — 99% purity, laboratory tested
60 capsules with Vitamin C + Organic Cocoa Powder



